This update includes key legal, regulatory, and industry updates are shaping the 340B landscape as 2026 begins.
Key Updates at a Glance
- Rebate Model Pilot remains paused; no Janaury 1 implementation
- HRSA confirms upfront 340B pricing continues
- HHS signals withdrawal of appeal
- IRA Medicare pricing now live and affecting margins
- Manufacturer ESP fragmentation increasing – new data submission platform “Truzo”
- No immediate operational changes required, but continued vigilance is needed
Courts Keep HRSA’s 340B Rebate Model Pilot on Hold
The 340B rebate model remains paused and did not go into effect on January 1, 2026, following a series of federal court rulings that blocked implementation. In early January, a federal appeals court denied the government’s emergency request to lift the injunction, leaving the pilot on hold nationwide while litigation and administrative review continue.
How the Block Took Effect
On December 29, 2025, a federal judge in the U.S. District Court for the District of Maine issued a nationwide injunction halting implementation of the rebate pilot. The court found that HRSA’s administrative record likely failed to adequately consider the operational and financial impact of a rebate-based pricing model on 340B hospitals.
Following the ruling, the Department of Health and Human Services (HHS) asked both the district court and the U.S. Court of Appeals for the First Circuit to allow the pilot to proceed during the appeals process. Both requests were denied.
On January 7, 2026, a three-judge panel of the First Circuit unanimously declined to lift the injunction, concluding that HHS had not shown it was likely to succeed on the merits of its appeal. The court emphasized that maintaining the injunction preserves the long-standing status quo of the 340B program. Recent filings indicate that HHS may no longer pursue appellate relief—shifting the outlook toward administrative reconsideration rather than near-term implementation.
What This Means for Covered Entities
Because the injunction remains in place, the rebate model did not go into effect on January 1. Manufacturers approved for the pilot must continue providing covered outpatient drugs at the 340B ceiling price through traditional upfront discounts.
How Long Could the Pause Last?
The injunction is temporary, but its duration remains uncertain. Recent developments suggest the pause may continue for an extended period while HRSA reassesses its approach. There is no defined end date unless the court takes further action or HRSA completes a revised administrative process.
What to Expect Next
With HHS signaling plans to withdraw its appeal, the pause of the rebate pilot is no longer tied to a defined appellate timeline. Instead, next steps are likely to occur through an administrative process, as HRSA reassesses the model considering the courts’ guidance. Any future implementation, whether of the current pilot or a revised approach, would require additional agency action, new guidance, and revised timelines. This process could materially delay or alter the structure of any rebate-based model.
For now, Covered Entities should continue operating under existing 340B frameworks and plan for ongoing uncertainty rather than a near-term resolution.
HRSA Confirms Ongoing 340B Operations During Rebate Pilot Pause
Following the court-ordered pause of HRSA’s 340B Rebate Model Pilot, recent developments suggest a potential shift in the federal government’s approach to the litigation, even as operational expectations for Covered Entities remain unchanged.
HHS Indicates Plans to Withdraw Appeal and Reconsider Approvals
In a January 12, 2026 filing with the U.S. Court of Appeals for the First Circuit, the Department of Health and Human Services (HHS) indicated it plans to withdraw its appeal of the order blocking the rebate pilot and return the approved rebate plans to HRSA for reconsideration. HHS stated it is engaged in discussions with the hospital groups that challenged the pilot and that both sides anticipate dismissing the appeal.
The filing follows multiple court rulings that found HHS likely failed to comply with administrative law requirements and did not adequately address the potential financial impact of a rebate-based pricing model on hospitals. If the appeal is dismissed, HRSA would need to reassess the approvals in light of the courts’ guidance before any future implementation could occur.
While the injunction remains in place, HRSA does not have authority to implement the rebate model. As a result, no rebate-based purchasing, reporting, or reconciliation processes are currently permitted.
Upfront 340B Pricing Remains Required
HRSA’s Office of Pharmacy Affairs has clarified that manufacturers must continue offering covered outpatient drugs at the 340B ceiling price as an upfront discount. Covered Entities are not required to purchase drugs at wholesale acquisition cost (WAC), submit rebate claims, or engage in Beacon platform reporting while the pilot is paused.
This guidance reinforces consistency across existing purchasing and replenishment workflows and helps limit confusion among wholesalers, pharmacy partners, and third-party administrators that had been preparing for a potential January 1 transition.
Audit Expectations Are Unchanged
Despite the policy uncertainty surrounding the rebate pilot, HRSA’s audit and program integrity activities continue without interruption. Recent audits continue to focus on familiar areas, including:
- Diversion and duplicate discount prevention
- Contract pharmacy oversight
- Documentation and record retention
- Internal controls across pharmacy, billing, and finance functions
Periods of transition or uncertainty can increase audit risk if roles, documentation standards, or processes become unclear. Maintaining stable workflows and clear internal ownership remains essential.
Why This Matters for Covered Entities
While HHS has signaled a possible shift away from defending the current rebate model, this development does not change current compliance obligations. Covered Entities should continue operating under existing 340B requirements and avoid making premature operational changes based on potential future scenarios.
At the same time, the broader environment—including overlapping federal drug pricing initiatives, increased manufacturer oversight, and evolving data infrastructure—underscores the importance of coordination across pharmacy, compliance, finance, and operational teams.
Click here for more information on the Rebate Model pause, and what would be necessary from HRSA/HHS to see this model return.
IRA Medicare Drug Pricing Moves Forward in 2026
Separate from the 340B rebate pilot, January 1, 2026 marked the first year of implementation for Medicare drug pricing negotiated under the Inflation Reduction Act (IRA). Unlike the rebate pilot, these changes moved forward as scheduled and are now being operationalized across the Medicare Part D program.
While the IRA does not directly amend the 340B statute, its implementation is expected to have material downstream effects on 340B operations, particularly for Covered Entities that dispense or administer drugs subject to Medicare price negotiation.
What the IRA Changes—and Why It Matters for 340B
Under the IRA, Medicare has established a Maximum Fair Price (MFP) for a defined set of high-spend Part D drugs. Manufacturers are required to make those prices available through Medicare-specific channels beginning in 2026.
Although MFPs do not replace 340B ceiling prices, they introduce a new federally mandated pricing construct that exists alongside 340B, Medicaid, and commercial pricing. For Covered Entities, this creates additional complexity in understanding how negotiated Medicare prices interact with existing purchasing, billing, and reimbursement workflows.
Overlapping Drug Lists and Policy Timelines Increase Complexity
Several drugs selected for Medicare price negotiation were also included in HRSA’s proposed 340B rebate pilot. Even with the rebate pilot paused, the overlap reinforces that IRA implementation and 340B policy are increasingly intertwined, particularly for high-cost outpatient drugs.
As manufacturers and PBMs operationalize MFP requirements, Covered Entities may encounter new terminology, contract language, claims edits, or data requests tied specifically to Medicare pricing. These changes may surface through pharmacy systems, revenue cycle processes, or communications from external partners.
For example, if a drug is purchased at a 340B price of $600 and Medicare previously reimbursed $1,000, the savings generated on that claim would be $400. If IRA pricing reduces Medicare reimbursement to $800, the drug is still purchased at $600, but the savings generated on that claim would decrease to $200.
How IRA Pricing Could Affect 340B Savings in Practice
Consider a high-cost outpatient drug that is both subject to Medicare’s negotiated Maximum Fair Price under the IRA and purchased by a Covered Entity at the 340B ceiling price. Under a traditional 340B arrangement, savings are generated based on the difference between the 340B acquisition cost and the payer reimbursement, including Medicare Part D reimbursement.
With the IRA in effect, Medicare Part D plans will reimburse based on the negotiated MFP for that drug. If the MFP is lower than prior Medicare reimbursement levels, the overall reimbursement associated with that claim may decrease. While the Covered Entity would still acquire the drug at the 340B price, the spread between acquisition cost and reimbursement could narrow, reducing the savings generated on Medicare Part D claims.
For contract pharmacies, this dynamic may appear as lower reimbursement amounts on Medicare claims for negotiated drugs, which could affect dispensing margins or fee arrangements tied to Medicare volume. Importantly, these changes stem from Medicare pricing policy, not from any change to 340B pricing requirements. The impact will vary by drug, payer mix, and contract structure, and does not affect non-Medicare claims.
Operational Considerations for Covered Entities
IRA-related changes may indirectly affect Medicare reimbursement levels, contract pharmacy claim processing for Part D beneficiaries, manufacturer pricing strategies, and internal assumptions about drug margin stability. These developments should not be assumed to apply to 340B purchasing or compliance without confirmation. However, they may influence how certain drugs perform financially across payer mixes, which can affect broader program planning.
Clear internal coordination between pharmacy, finance, revenue cycle, and compliance teams will be increasingly important to distinguish IRA-driven changes from 340B requirements and avoid unnecessary workflow adjustments or pricing disputes.
Why This Matters Heading Into 2026
Unlike the rebate pilot, IRA drug pricing is fully implemented and ongoing. As additional drugs are selected for negotiation in future years, the interaction between Medicare pricing policy and 340B operations is likely to become more pronounced.
For Covered Entities, the challenge will not be replacing 340B pricing but managing an increasingly layered pricing environment in which multiple federal programs influence drug economics at the same time.
Industry Funding Data Highlights Continued Pressure on 340B Policy
Newly released IRS tax filings provide additional context on the scale and structure of advocacy efforts shaping federal and state health care policy, including ongoing debates surrounding the 340B Drug Pricing Program.
The filings, which reflect 2024 revenue and spending reported by major pharmaceutical and provider trade associations, underscore the significant resources being directed toward policy development, research, and public advocacy related to drug pricing and provider reimbursement.
Pharmaceutical and Provider Groups Play Distinct Roles
Among the organizations reviewed, Pharmaceutical Research and Manufacturers of America (PhRMA) reported the highest total revenue in 2024. The filings also show that PhRMA and other drug industry groups provided funding to outside organizations that have taken positions critical of aspects of the 340B program, including contract pharmacy access and rebate-based pricing models.
At the same time, hospital and provider associations—including the American Hospital Association, 340B Health, and America’s Essential Hospitals—reported continued advocacy activity focused on defending the structure of the 340B program and opposing rebate-based approaches.
Why This Context Matters
While specific policy initiatives may be paused or reconsidered, these filings highlight that broader advocacy efforts related to 340B remain active and well-resourced across the health care sector. Competing perspectives continue to influence litigation, legislation, and regulatory strategy at both the federal and state levels. For Covered Entities, this context helps explain why 340B policy debates persist even during periods of operational stability and why future proposals—whether legislative, regulatory, or litigation-driven—are likely to continue emerging.
Rural Pharmacy Access in a Changing Policy Landscape
Recent legal and policy developments continue to highlight the importance of contract pharmacy access for rural providers and the patients they serve. More than half of rural hospitals do not operate an on-site outpatient pharmacy, and over 60% of rural counties are considered pharmacy deserts or low-access areas, making contract pharmacy arrangements a critical mechanism for maintaining outpatient medication access in many communities.
As state-level contract pharmacy protections expand and litigation continues to shape manufacturer behavior, the implications for rural access remain an important consideration. While day-to-day operations may not change immediately, shifts in the legal landscape can have downstream effects on how rural providers sustain pharmacy services, manage patient access, and support continuity of care over time.
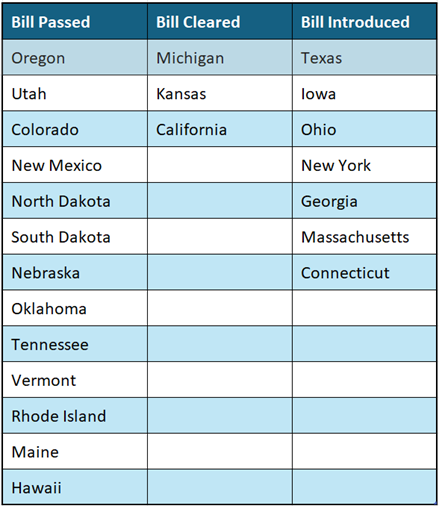
Current Status on State Bills and Laws that Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates since last update
Truzo Platform Gains Manufacturer Adoption
In recent months, some manufacturers have begun transitioning select 340B Electronic Submission Platform (ESP) functions away from HRSA-supported infrastructure and onto third-party platforms. These changes reflect manufacturer-specific decisions about how 340B designations and claims data are managed, rather than new requirements issued by HRSA.
Manufacturers including Puma Biotechnology, Argenx, and Amgen have communicated plans to utilize Truzo, a third-party platform operated by Kalderos, for certain 340B ESP–related processes. Adoption varies by manufacturer and product and is occurring independently of HRSA’s paused rebate pilot.
What Is Changing
While HRSA’s ESP remains in place, some manufacturers are choosing to manage specific 340B administrative functions through Truzo, including:
- Covered Entity and contract pharmacy designations
- Claims data submission and validation
- Manufacturer oversight and reconciliation processes
These activities are managed at the manufacturer level and do not automatically apply across all products or Covered Entities.
Why This Matters
Even as HRSA’s rebate pilot remains on hold, manufacturer movement toward third-party ESP infrastructure reflects a broader trend toward increased data oversight and manufacturer-managed controls within the 340B program. Platform fragmentation increases the importance of understanding where claims data is expected to be submitted, and which platform applies to specific manufacturers and NDCs.
What Covered Entities Should Do
Covered Entities that purchase drugs from manufacturers utilizing Truzo should ensure they are registered on the appropriate platform and prepared to submit claims data as required by the manufacturer. Claims for affected NDCs submitted through HRSA’s 340B ESP will not be accepted, as HRSA’s ESP and Truzo do not cross-communicate. In these cases, NDCs associated with participating manufacturers may be removed from HRSA’s ESP environment.
Registration is not necessary if a Covered Entity does not purchase or dispense affected NDCs, but awareness of platform requirements and manufacturer-specific shifts remains important as manufacturers continue to adjust how 340B data and designations are managed.

