Key Updates at a Glance
- HRSA rebate model advances to public comment (Docket HRSA-2026-03042; comments due April 20)
- IRA Medicare negotiated pricing now flowing through 2026 claims, margin compression emerging
- Eli Lilly Implements Expanded 340B Claims Reporting for In-House Dispensing
- Contract pharmacy litigation and state-level enforcement activity continue
- Manufacturer data oversight and audit expectations increasing
340B Rebate Proposal Back in Play – HRSA Opens Comment Period
The White House Office of Management and Budget (OMB) has completed its initial review of HHS’s new 340B Drug Pricing Program Manufacturer Rebate Models pre-rule. While the proposal text has not yet been released and no publication timeline has been announced, the brief five-day review window signals the administration is moving quickly to recalibrate its approach following last year’s halted rebate pilot. OMB’s determination of “consistent with change” means the concept may proceed, subject to revisions requested during review. Details of those revisions have not been disclosed.
HRSA Requests Stakeholder Feedback
Just two days later, the Health Resources and Services Administration (HRSA) released a 14-page Request for Information (RFI) seeking feedback from stakeholders on how a 340B rebate system could be structured—if pursued.
The RFI, scheduled for publication in the Federal Register on February 17, requests input from hospitals, manufacturers, pharmacies, wholesalers, and state Medicaid agencies on:
- Administrative and cash-flow impacts of shifting from upfront discounts to rebates
- Safeguards to prevent duplicate discounts
- Data and transparency improvements for program oversight
Public comments must be submitted by March 19, 2026. (Docket ID: HRSA-2026-03042)
HRSA describes this effort as a “methodical and deliberate” reset, emphasizing that stakeholder input will inform whether the agency develops a new pilot or initiates formal rulemaking.
Key Dates:
- February 17: RFI published in the Federal Register
- March 19: Deadline to submit comments
- Spring 2026: HRSA expected to review feedback and decide whether to move forward with developing a new rebate pilot
If HRSA proceeds, it must issue a new notice, open another public comment window, and provide ample lead time before implementation, aligning with the agency’s new procedural commitments.
What It Means for 340B Stakeholders
The administration’s recent actions indicate continued evaluation of a potential 340B rebate model, though no formal proposal has been issued. At this stage, the RFI functions primarily as an information-gathering step and provides insight into the agency’s current areas of focus. Any substantive policy change would require additional notice and procedural steps before implementation. In the interim, organizations can monitor developments and assess potential operational or compliance implications as further details become available.
To review the RFI on the Federal Register, click here.
340B Margins Contract as Medicare Negotiated Prices Take Effect
January 1 marked the implementation of Medicare’s negotiated drug pricing under the Inflation Reduction Act (IRA), applying Maximum Fair Prices (MFPs) to the first 10 high-cost Part D drugs. After one month in 2026, Covered Entities (CEs) are seeing the real-world reimbursement effects reflected directly in claim payments.
Early Financial Signals
340B ceiling prices have not changed. However, Medicare Part D reimbursement for the negotiated drugs is now lower under the new Maximum Fair Price (MFP) structure. That means the difference between 340B acquisition cost and Medicare payment — the margin that supports program operations — has narrowed.
- Medicare Part D margins on negotiated drugs have declined significantly, exceeding the modest to mid-range impact many early industry analyses projected.
- High-volume drugs such as Eliquis, Xarelto, Jardiance, and Farxiga are showing noticeable margin compression.
- Finance and pharmacy teams are recalibrating margin expectations as lower Medicare reimbursement levels become clear.
The result is a clear narrowing of Medicare Part D margins on negotiated drugs, affecting both Covered Entity program revenue and contract pharmacy economics.
Why This Is Happening
The IRA requires manufacturers to offer negotiated Maximum Fair Prices within Medicare Part D beginning in 2026. Although 340B purchase pricing remains intact, the reimbursement side of the equation — particularly for Medicare beneficiaries — has effectively reset at lower levels.
Put simply, 340B savings are driven by the difference between acquisition cost and reimbursement. When Medicare reimbursement declines but acquisition costs remain the same, the spread narrows. For organizations and pharmacies with significant Medicare Part D volume, even incremental per-claim reductions can translate into meaningful financial impact. Non-Medicare claims (commercial, Medicaid, uninsured) are not experiencing the same immediate compression, making Medicare volume the primary driver of current margin contraction.
What to Expect Next
- Expanded negotiations: Additional drugs are scheduled for negotiation in 2027 and beyond, potentially extending margin pressure.
- Independent pharmacy negotiations: As Medicare reimbursement tightens, contract pharmacies may seek to renegotiate dispensing fees or revenue-share arrangements to offset reduced Part D margins.
- More complex tracking: Distinguishing IRA-driven reimbursement changes from 340B pricing or compliance issues will require tighter coordination among pharmacy, revenue cycle, finance, and compliance teams.
- Increased manufacturer oversight: Covered Entities should expect greater scrutiny around duplicate discounts, data accuracy, and documentation as pricing pressure increases.
Eli Lilly Expands 340B Claims Reporting to In-House Pharmacies and Medical Claims Data
Beginning February 1, 2026, covered entities must submit claims-level data for Eli Lilly products through the 340B ESP platform. Lilly has indicated that failure to provide “timely, complete, and accurate” data could result in loss of access to 340B pricing until reporting requirements are met. Lilly has stated the purpose is to prevent duplicate discounts and strengthen program integrity. This marks a significant shift in the 340B compliance environment.
How We Got Here: Key Timeline
- 2020: Lilly becomes the first manufacturer to restrict 340B contract pharmacy distribution, triggering widespread industry litigation and policy disputes.
- 2023–2024: Federal appellate court decisions clarify that manufacturers may impose certain distribution conditions, reshaping the legal landscape.
- 2025: Courts block manufacturer rebate models absent federal approval.
- February 1, 2026: Lilly expands oversight by requiring claims-level reporting for drugs dispensed through in-house pharmacies.
Until now, most manufacturer actions targeted contract pharmacy arrangements. In-house dispensing was generally viewed as more operationally stable. This policy narrows that distinction.
What Is Changing Operationally
Under the expanded policy:
- Claims-level dispensing data must be submitted for all Lilly products. (Labeler codes 00002, 00777, 66733)
- All 340B dispenses, regardless of channel
- Reporting flows through the 340B ESP platform.
- Data completeness and timeliness are explicitly tied to continued 340B pricing access.
Provider groups, including the American Hospital Association and 340B Health, have raised concerns that conditioning statutory 340B pricing on expanded reporting requirements increases administrative burden and may exceed manufacturer authority under the 340B statute.
This development reflects a broader evolution in 340B oversight, shifting attention from distribution models to claim-level validation and data transparency. Compliance expectations are expanding beyond eligibility and diversion prevention, and in-house pharmacy models may now face scrutiny comparable to contract pharmacy arrangements.
For many Covered Entities, the operational impact is less about pricing mechanics and more about infrastructure — IT systems, data extraction capabilities, audit controls, and staffing resources.
What to Expect Next
Covered Entities should take this as a prompt to review their data capabilities and internal reporting workflows. Understanding how claim-level dispensing data is captured, reconciled, and validated will be critical if expanded reporting becomes more common. Organizations should also carefully evaluate manufacturer communications and monitor regulatory response. As oversight shifts toward data transparency, readiness will position entities to comply, respond, or advocate effectively as the policy landscape continues to evolve.
To read more on this new policy, you can click here.
Contract Pharmacy Landscape Updates
State legislatures, federal courts, and manufacturers all moved in the last 30 days — reinforcing that contract pharmacy remains one of the most dynamic areas of 340B policy.
State Legislative Movement
Activity continues across multiple states:
- Virginia – Access legislation advancing under new gubernatorial leadership.
- Washington – Bills progressing that pair access protections with added reporting requirements.
- Michigan & Iowa – Measures advancing with bipartisan support.
- Arizona – Newly introduced bill would prohibit manufacturer-imposed restrictions not required under federal law.
- Advocacy activity intensified in Chicago, urging movement on stalled legislation.
Trend: States are increasingly asserting authority over contract pharmacy access, with some adding structured reporting components.
Litigation Developments
Recent court activity includes:
- Federal court decisions in Maine have declined to block enforcement of the state’s law.
- Ongoing challenges in North Dakota and Oregon.
- Appeals activity continues in several jurisdictions.
Pattern: District courts have generally been reluctant to halt state laws while litigation proceeds.
Manufacturer Policy Updates
Several manufacturers revised policies in recent weeks:
- Pfizer – New attestation requirements and product adjustments.
- Sumitomo Pharma America – Expanded state exemptions with continued data conditions.
- Sandoz – Additional products added to restriction frameworks.
Even in protected states, manufacturer-specific compliance requirements remain fluid.
What to Watch Next
- Floor votes in key states
- Summary judgment rulings in active litigation
- Further manufacturer refinements
- Expansion of “access + reporting” legislative models
Contract pharmacy access is increasingly shaped at the state level while manufacturers continue to refine restriction frameworks. For covered entities, this translates to ongoing operational monitoring, state-by-state compliance variation, and potential financial impact tied to evolving access models.
What We Heard at the 340B Coalition Winter Conference
Our team at ACI 340B Management recently attended the annual conference hosted by 340B Health in San Diego, where compliance, pharmacy, and policy leaders from across the country gathered to discuss the rapidly evolving 340B landscape.
Advocacy for the 340B program was a central theme throughout the conference. With ongoing manufacturer policy shifts, litigation developments, and increasing data oversight expectations, there was strong emphasis on protecting the integrity and intent of the program. The message was clear: collaboration, education, and proactive engagement with policymakers will be critical to ensuring 340B remains sustainable for the safety-net providers and patients it supports.
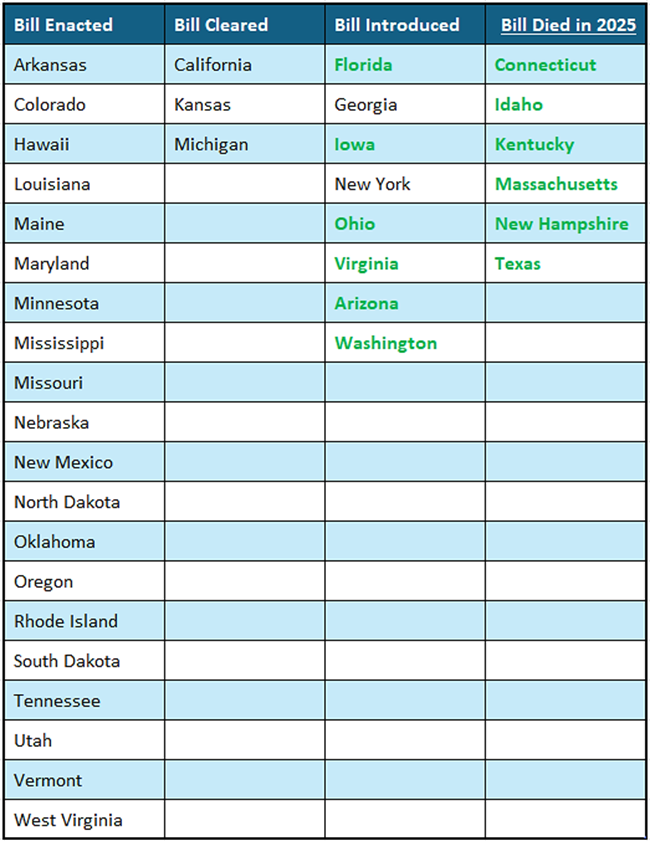
Current Status on State Bills and Laws That Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates.
Washington Update: 340B Returns to the Hearing Room
The 340B program resurfaced during a Feb. 11 hearing before the U.S. House Energy and Commerce Health Subcommittee focused on prescription drug supply chain costs.
Although the hearing was not dedicated to 340B, pharmaceutical industry leaders used the platform to renew calls for greater scrutiny of the program — reinforcing that 340B remains part of the broader federal drug pricing conversation.
Industry Leaders Renew Reform Narrative
During testimony, Lori Reilly, Chief Operating Officer of Pharmaceutical Research and Manufacturers of America (PhRMA), described 340B as one of the fastest-growing federal drug programs and urged lawmakers to examine hospital use of discounted medicines. She pointed to transparency concerns and called for structural reforms, reiterating industry support for rebate-based compliance approaches and legislative proposals aimed at tightening hospital participation standards.
In written testimony, John Crowley, CEO of the Biotechnology Innovation Organization (BIO), echoed concerns about hospital pricing practices and program growth, advocating for increased accountability measures.
Notably, lawmakers did not directly question witnesses on 340B during the hearing, and no immediate legislative action resulted.
Provider Pushback and Policy Signals
Ahead of the hearing, the American Hospital Association (AHA) defended the program as a critical safety-net tool that allows eligible providers to stretch limited resources and expand access to care. Hospital groups continue to argue that proposed structural changes could create administrative burden and financial instability for participating entities.
While the hearing itself did not produce concrete policy movement, it reinforces a clear trend: 340B remains a focal point in federal drug pricing reform discussions. Continued industry messaging around markups, transparency, and program growth suggests sustained pressure in 2026, even absent immediate legislative changes.
For Covered Entities, the signal is continued scrutiny rather than immediate operational disruption. However, awareness of the policy climate remains essential as reform conversations evolve.
Engagement Matters
As federal scrutiny continues, proactive engagement is increasingly important. Covered Entities and stakeholders should consider educating their federal and state representatives about how 340B supports patient access, community services, and financial sustainability. Clear, data-driven communication today can help shape how policymakers understand the real-world impact of potential reforms tomorrow. As 2026 unfolds, the 340B program is facing simultaneous financial, operational, and legislative pressure points, making proactive strategy more important than ever.

