Key 340B Updates at a Glance:
- State contract pharmacy laws hold in court, with wins in Tennessee, Colorado, and Minnesota. Kansas faces a June 30 deadline shaping near-term enforcement direction.
- Eli Lilly, Novo Nordisk, Bristol Myers Squibb, and AstraZeneca expand in-house pharmacy claims reporting, extending 340B data requirements beyond contract pharmacy.
- PDAB activity expands, with four states now holding UPL authority. Implementation remains unsettled, with varied approaches to 340B treatment.
- Amgen and GSK update manufacturer policies, impacting contract pharmacy exemptions and specialty pharmacy network designations.
- Idaho advances reporting simplification legislation, while Community Voices for 340B launches a Litigation Defense Fund for contract pharmacy challenges.
- HRSA rebate model RFI closes April 20. Form letter activity reported in docket; provider groups, including AHA, urge increased participation.
State Contract Pharmacy Laws: Major Wins, One Setback
State-level 340B contract pharmacy litigation produced a wave of provider wins this period, even as manufacturer challenges continued to mount. Washington became the 22nd state to enact a contract pharmacy access law when Gov. Bob Ferguson signed S.B. 5981 on March 25, the first state to do so in 2026, and notably the first to pair contract pharmacy protections with mandatory reporting requirements for both providers and manufacturers.
The law takes effect June 10 and immediately drew lawsuits from AbbVie and Novartis, who filed suit the same day it was signed.
Provider Wins Stack Up in Court
Courts across the country continued to side with states and providers this period:
- Tennessee: A federal judge dismissed both AbbVie’s and PhRMA’s separate lawsuits challenging the state’s contract pharmacy law within a three-week span. The court specifically found that Tennessee’s prohibition on manufacturers requiring claims data not mandated under federal law was consistent with federal statute, a ruling with direct operational significance given Lilly’s and Novo Nordisk’s current in-house pharmacy data demands. Lilly has already exempted providers in Tennessee and 11 other states with similar laws from its in-house reporting requirement.
- Colorado: A third federal judge denied PhRMA’s request to block the state’s contract pharmacy law, marking the third consecutive ruling in favor of the statute from three different Colorado federal judges.
- Minnesota: A state appeals court upheld the state’s 2024 contract pharmacy statute, protecting access for more than 200 covered entities and roughly 350,000 patients. A prior conflicting federal ruling means further legal developments there should be watched closely.
- New York: The Senate Finance Committee unanimously advanced S. 1913, the “340B Prescription Drug Anti-Discrimination Act,” which would bar both manufacturers and PBMs from restricting or discriminating against 340B contract pharmacy arrangements. A full Senate floor vote is pending.
Kansas Setback Puts June 30 Deadline in Play
The one significant legislative setback this period: Kansas House Speaker Dan Hawkins abruptly ended the chamber’s session on March 19 to block a floor vote on S.B. 284, a bipartisan contract pharmacy access bill that had passed the Senate 34-6 and was broadly expected to pass the House. The maneuver drew sharp criticism from state providers and lawmakers. The existing Kansas law protecting contract pharmacy access expires June 30, 2026, and the legislature is officially scheduled to adjourn April 10, leaving a narrow window for action.
What This Means for CEs
Covered entities in states with enacted laws are in a stronger position but must remain prepared for ongoing litigation uncertainty. CEs in Kansas should assess contingency plans now given the June 30 expiration. CEs in states where legislation is still pending — including New York, Iowa, Georgia, Florida, Ohio, and Arizona — should stay engaged with state advocacy efforts, as 2026 remains an active year at the state level.
In-House Pharmacy Reporting Requirements Continue to Expand
A 340B Administrative Dispute Resolution (ADR) panel recently rejected a hospital challenge to a drugmaker’s 340B contract pharmacy restrictions, marking the sixth consecutive time an ADR panel has sided with manufacturers under the updated process.
The latest ruling dismissed a petition from Wisconsin-based St. Croix Regional Medical Center against Danish drugmaker Novo Nordisk’s contract pharmacy limitations, citing a January 2023 Philadelphia federal appeals court decision that upheld similar restrictions. The panel found “no overcharge violation” under the 340B statute.
The decision does not address Novo Nordisk’s newly announced requirement for covered entities (CEs) to submit claims data for in-house pharmacy dispenses to access 340B pricing, a policy set to take effect April 1 that providers argued is unlawful and that the company says enhances transparency.
Background on the ADR Process
Eli Lilly, Novo Nordisk, and now AstraZeneca and Bristol Myers Squibb (BMS) have all moved to expand their 340B data reporting requirements beyond contract pharmacies to include all in-house pharmacy dispenses. Lilly requires covered entities to submit claims data within 45 days of dispense, while Novo Nordisk’s similar policy requiring both pharmacy and medical claims data for every 340B in-house dispense took effect April 1.
Failure to submit complete or timely data under either policy could result in suspension of 340B pricing. AstraZeneca announced April 13 that beginning May 1, it will require claims data on all 340B purchases across 19 of its products, including Farxiga and Symbicort, applying the policy across all states.
Effective May 1, 2026, BMS will require covered entities to submit claim-level data elements for all 340B utilization of certain covered outpatient drugs, covering dispenses and administrations from both entity-owned pharmacies and designated contract pharmacies.
Legal Status Remains Unsettled
These policies are actively contested, and the legal and regulatory picture remains murky for covered entities:
- HRSA has not formally weighed in on the legality of these requirements. Absent federal guidance, the risk of non-compliance, including suspension of 340B pricing, remains real regardless of provider objections.
- State law may offer some protection. CEs in states with claims data prohibition laws, including Tennessee and potentially others, may be exempt from certain requirements. Lilly has already carved out providers in 11 such states from its in-house reporting policy; Novo Nordisk has not made similar exemptions. AstraZeneca and BMS similarly announced their policies will apply across all states, with no exemptions noted.
- ADR has offered little relief. A recent panel ruled against Wisconsin-based St. Croix Regional Medical Center in its challenge to Novo Nordisk’s contract pharmacy restrictions, marking the sixth consecutive ADR ruling in favor of manufacturers. CEs should not rely on the ADR process as a primary strategy for contesting these policies.
Operational and Financial Pressure is Real
The compliance burden of these new requirements is significant, particularly for under-resourced providers:
- New EMR integrations, additional staff time, or third-party vendor support may be required to handle increased data volume.
- Smaller clinics and FQHCs face the greatest exposure, as they already carry significant reporting obligations and limited administrative capacity.
- Data collected to satisfy manufacturer requirements may later overlap with potential HRSA reporting standards if a federal rebate model moves forward — creating dual-use compliance considerations and raising strategic questions about data governance across both obligations simultaneously.
What This Means for CEs
Assess your reporting capabilities immediately for all four manufacturers. Determine whether your state’s law provides any protection against claims data mandates.
Consider third-party support if internal systems cannot handle the volume. Document your compliance processes carefully, and monitor for other manufacturers that may follow with similar in-house reporting policies.
Evaluating the Impact of PDABs on 340B Savings & Sustainability
State Prescription Drug Affordability Boards (PDAB) are drawing less attention than contract pharmacy battles, but for covered entities, they represent a meaningful and evolving financial consideration. At least 11 states have created a PDAB or similar body since 2019, with Colorado, Maryland, Minnesota, and Washington holding UPL authority.
A key concern for covered entities is the potential for margin compression: a UPL lowers the reimbursement ceiling on a given drug, compressing the margin between what a CE pays at the 340B acquisition cost and what it receives in reimbursement.
Where Things Stand by State
As implementation progresses, variation across states is creating a fragmented policy landscape with different implications for 340B programs. The four states with UPL authority are at different stages of implementation, and each presents distinct considerations for CEs:
- Colorado set a $600-per-unit UPL on Enbrel effective January 1, 2027. Amgen has filed a legal challenge arguing that the limit is unconstitutional, citing many of the same arguments manufacturers have used against state contract pharmacy access laws.
- Maryland is developing UPL frameworks for Jardiance and Farxiga. The state enacted a law intended to carve out 340B drugs from UPL application, but experts have noted it lacks the infrastructure to distinguish 340B from non-340B claims at the transaction level, raising operational questions about how effectively the carveout can be implemented. The Maryland PDAB also announced in March that it may require covered entities and manufacturers to report 340B program data as part of a state-level study, adding another potential reporting layer on top of existing obligations.
- Minnesota is in early design stages and has not yet addressed 340B carveouts.
- Washington’s PDAB has not yet publicly addressed 340B program implications.
The Carveout Dilemma
Whether or not a state attempts to protect 340B drugs through a carveout, covered entities may face financial tradeoffs under either approach. If 340B drugs are included in a UPL, provider savings are directly reduced. If 340B drugs are carved out, payers may be incentivized to steer patients toward non-340B alternatives where their costs are lower, potentially eliminating savings from those drugs altogether.
There is no clearly established approach under current PDAB frameworks, and states are still working through the implementation challenges without clear answers.
What This Means for CEs
If you operate in Colorado, Maryland, Minnesota, or Washington, monitor PDAB activity closely and engage in rulemaking processes where possible, including through state and national associations.
Proactively model how a UPL on high-revenue drugs could affect your program’s financial performance and ensure 340B program impacts are represented in affordability board deliberations.
Recent Manufacturer Policy Changes Affecting 340B Covered Entities
As the broader 340B legal and policy landscape dominates headlines, manufacturer-level policy changes continue to create real operational exposure for covered entities. These shifts, often announced quietly through third-party vendor portals or manufacturer websites, can affect which pharmacies a CE can use, which drugs are accessible at 340B pricing, and whether a CE’s current designations remain compliant.
Staying current on these changes is not optional; a missed update can result in lost 340B pricing with little warning.
Amgen and GSK: What Changed and What It Means
Two manufacturers made notable policy adjustments this period that CEs should be aware of:
Amgen revised its contract pharmacy policy to exempt Oregon providers following the state’s enactment of a contract pharmacy access law, while simultaneously removing its prior exemption for New Mexico providers.
CEs in New Mexico who had been operating under that exemption need to ensure their contract pharmacy arrangements are now compliant with Amgen’s standard policy, which generally limits 340B shipments to an in-house or single contract pharmacy when claims data are submitted.
GSK expanded its specialty pharmacy network for Exdensur, a treatment for severe asthma, adding Caremark, CareMed, and Walgreens Specialty Pharmacy to its limited network.
CEs without an in-house pharmacy capable of dispensing the drug must designate a single contract pharmacy, and that designation must be within GSK’s limited network. Exdensur is one of six GSK drugs subject to limited pharmacy network restrictions, and CEs should confirm their designations are aligned for each one.
Building a Process Around Policy Monitoring
The Amgen and GSK updates reflect a broader pattern: manufacturer policies are not static, and the consequences of missing a change fall entirely on the covered entity. Most policy updates are communicated through 340B ESP or manufacturer websites rather than direct outreach, meaning CEs without a systematic monitoring process are at risk of falling out of compliance without realizing it.
As more manufacturers expand restrictions and modify exemptions in response to the evolving state law landscape, the frequency of these changes is only likely to increase.
A Growing Pattern of Fragmented Manufacturer Policy
These updates are not isolated events. Across the manufacturer landscape, 340B policies are increasingly diverging by state, drug class, and distribution channel. As manufacturers respond to state contract pharmacy laws and broader pricing pressure, covered entities are seeing a shift from uniform national policies to highly variable, drug-specific restrictions. This fragmentation increases the operational burden on covered entities and raises the risk of unintentional non-compliance.
What This Means for CEs
Audit your current contract pharmacy designations and specialty pharmacy assignments across all restricted manufacturers — not just Amgen and GSK. Confirm that any state-based exemptions you are operating under are still active, particularly if your state has seen recent legal or legislative developments.
Establish a routine monitoring process for manufacturer policy updates through 340B ESP and manufacturer websites and ensure your third-party administrator or compliance team is flagging changes in real time.
Idaho Reporting Update and the New 340B Litigation Defense Fund
Idaho’s Senate State Affairs Committee introduced S.B. 1390, which would narrow the state’s existing 340B provider reporting law to focus specifically on contract pharmacy acquisition costs and reimbursement — rather than all dispenses — while retaining the requirement to report how 340B savings are used. The move reflects pushback from state hospitals who argued the original law created significant administrative burden without clear patient benefit. Idaho’s first reports under the existing law were due April 1, so CEs in the state should ensure current compliance while the amendment works through the process.
Community Voices for 340B (CV340B) also launched a 340B Litigation Defense Fund in March to provide financial support to stakeholders defending contract pharmacy access laws against manufacturer lawsuits. The fund is accepting both contributions and applications, and applicants are not required to use specific legal counsel.
Idaho CEs should file April 1 reports as required and monitor S.B. 1390’s progress. Those interested in the CV340B Litigation Defense Fund can find more information and apply through the CV340B website.
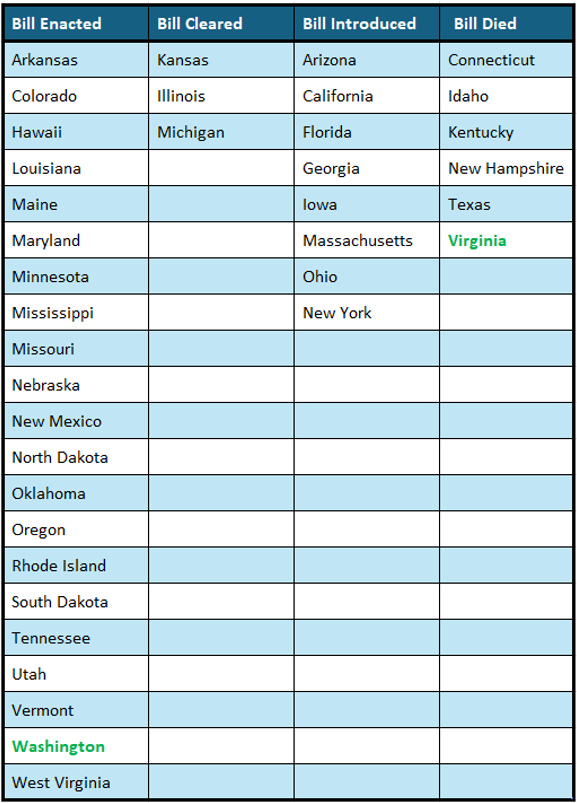
Current Status on State Bills and Laws that Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates since last update
Washington Update: The Rebate Model Fight Heads to Congress
Congressional opposition to a 340B rebate model is gaining momentum heading into appropriations season. A bipartisan group of 94 U.S. House members sent a March 27 letter calling for language in the FY 2027 HHS spending bill that would prohibit any appropriated funds from being used to implement a 340B rebate model — covering the now-halted original pilot, any new model HRSA is considering, and any future manufacturer-initiated proposals.
The letter was led by Reps. Matsui (D-CA), Johnson (R-SD), Dingell (D-MI), and Mann (R-KS), and signed by 79 Democrats and 15 Republicans.
HRSA RFI Comment Period is Ending
Patients Rising, a nonprofit with more than a dozen pharmaceutical company sponsors, was recently caught flooding the RFI comment pool with identical template submissions. Over half of the first 299 published comments were near-identical letters submitted in alphabetical order over a three-day period.
After 340B Report exposed the campaign, Patients Rising shut it down. The manufacturer perspective is being organized and submitted at volume — provider voices need to counter it with what form letters cannot offer: real program data and real patient impact. A July 2025 survey from 340B Health found that a rebate model could require the average 340B disproportionate share hospital to float an estimated $72.2 million to manufacturers annually.
Comments should speak to:
- The cash flow impact of purchasing drugs at full cost while awaiting rebate reimbursement
- The administrative burden of claims submission, reconciliation, and compliance infrastructure
- The specific patient services your 340B savings support that would be at risk
340B Under the Microscope on Capitol Hill
Beyond the rebate debate, the program is facing scrutiny on several fronts simultaneously:
- House E&C Hearing: The Health Subcommittee held a March 18 hearing where the subcommittee chair called the program “opaque” and a witness proposed replacing 340B with a safety-net grant program entirely. Provider groups responded with strong defenses.
- The ACCESS Act: An act which would significantly restrict hospital 340B eligibility, remains stalled with no new co-sponsors.
- False Claims Act: The 9th Circuit revived a whistleblower lawsuit against AbbVie, AstraZeneca, Novartis, and Sanofi alleging 340B overcharges, which could increase federal scrutiny of manufacturer pricing compliance.
What This Means for CEs
Submit comments before April 20 — individually, not just through your association. Contact your state hospital association and Congressional delegation directly. The window to shape this conversation is closing.
Check out our last edition of ACI Monthly here.

