This issue highlights new federal policy shifts affecting 340B, including CMS’s proposal to track 340B drugs in Medicare Part D and HRSA’s rebate model pilot. We also cover evolving compliance expectations and broader developments impacting patient access and program stability.
Policy Shift to Identify 340B Drugs in Medicare Part D
The Centers for Medicare & Medicaid Services (CMS) has proposed two new methodologies to identify 340B-discounted drugs billed to Medicare Part D. The proposed policy is part of the agency’s efforts to comply with provisions of the 2022 Inflation Reduction Act (IRA). The law requires drug manufacturers to pay rebates to Medicare when drug prices rise faster than inflation. However, 340B drugs are specifically excluded from these calculations, making it essential for CMS to clearly identify which claims are for 340B drugs, a step the agency has not taken until now.
Two New Tracking Methods: Cross-Referencing and Voluntary Reporting
CMS has proposed a dual-track strategy to identify claims. The first approach relies on cross-referencing existing provider and pharmacy National Provider Identifiers (NPIs) with known 340B contract relationships. If a prescriber or dispensing pharmacy is tied to a 340B covered entity, CMS would presume the associated drug claim to be 340B eligible. This passive methodology could allow CMS to scale oversight quickly using existing data, though questions remain about the accuracy of relying solely on presumed relationships.
The second proposal is more proactive: CMS would create a new 340B claims data repository that covered entities could begin using voluntarily in 2026. Participants would report five data elements for each Medicare Part D claim involving a 340B drug. This would include the prescription fill date, prescription reference number, fill number, the NPI of the dispensing pharmacy, and the 11-digit National Drug Code (NDC).
While CMS emphasizes that the 2026 reporting period is voluntary, the rule strongly suggests this could become mandatory in the future. CMS has also stated that the collected data will be confidential and will not be shared with drug manufacturers or Medicare Part D plan sponsors.
Laying the Groundwork for Broader Oversight
This proposal represents CMS’s first formal attempt to bring visibility into 340B activity under Medicare Part D, a segment of the program that has long lacked transparency due to limited reporting infrastructure. This new oversight could influence future enforcement on issues such as duplicate discounts, an area where CMS has thus far remained silent, despite increasing pressure from both drug manufacturers and providers.
What to Expect Next:
Covered entities need to assess their current capabilities to track and report 340B claims data at the level of detail CMS is requesting. Many providers currently determine 340B status retrospectively, which complicates reporting. Internal systems may need upgrades, and coordination between pharmacy teams, compliance officers, and IT departments will be critical.
Even those not planning to participate in the voluntary testing period should begin preparing systems in place for likely mandatory participation in 2027 or beyond. Stakeholders have until September 12, 2025, to submit comments to CMS. Given the technical complexity and potential financial impact of these changes, early engagement in the rulemaking process is critical.
HRSA’s New 340B Rebate Model Pilot Program
The Health Resources and Services Administration (HRSA) has announced the launch of a 340B Rebate Model Pilot Program, a voluntary initiative that may mark a shift in how some 340B ceiling prices are delivered. For covered entities, this new model could mean temporary changes in purchasing and reimbursement workflows—but also a formal opportunity to weigh in on the structure before it expands.
What’s Changing
Traditionally, the 340B Program provides upfront drug discounts at the point of sale. Under this new rebate model, participating drug manufacturers would charge full price upfront, and covered entities would receive a rebate afterward to reduce the net cost to the 340B ceiling price.
The pilot applies only to a select group of drugs, specifically those on the CMS Medicare Drug Price Negotiation Program (MDPNP) Selected Drug List for 2026, and only if the manufacturer chooses to participate and receives HRSA approval.
Key Dates for Covered Entities
- August 1, 2025: HRSA’s notice published in the Federal Register.
- September 15, 2025: Deadline for manufacturers to submit pilot participation plans.
- October 15, 2025: HRSA to announce approved participants.
- January 1, 2026: Pilot begins for approved drugs and manufacturers.
- Comment period open now: Covered entities can submit feedback on the pilot structure via regulations.gov until September 8, 2025.
What the Pilot Requires of Manufacturers
HRSA has built multiple safeguards into the pilot to reduce operational and financial disruption:
- No administrative costs of the rebate system can be passed on to covered entities.
- Covered entities will continue ordering through existing 340B wholesalers and distribution systems.
- Manufacturers must provide 60 days’ notice and clear instructions before implementation.
- Claims platforms must be secure, HIPAA-compliant, and allow entities to submit claims up to 45 days after dispense.
- Rebates must be issued or denied (with explanation) within 10 days of data submission.
- Manufacturers must provide real-time status reports on claim activity.
- Denials for concerns like diversion or duplicate discounts must follow proper channels, not arbitrary rejection.
What Covered Entities Should Watch
While the pilot is voluntary for manufacturers and applies only to a limited number of drugs, it could create temporary cash flow challenges, administrative work, and data coordination requirements for covered entities. However, HRSA has explicitly prohibited any cost shifting and will be monitoring manufacturer compliance closely.
Covered entities should be aware that:
- Rebates will only apply to specific drugs in the MDPNP list
- Not all manufacturers will participate
- Any implementation must follow HRSA’s pre-approval process
- Covered entities can raise concerns directly to HRSA during the pilot
Significance of HRSA Pilot
This is the first time HRSA has formally opened the door to a rebate-based 340B model, a proposal that previously generated concern among covered entities due to financial and administrative risk. With this pilot, HRSA is testing whether rebates can be implemented without undermining access or adding undue burden to safety-net providers.
Covered entities now have a critical opportunity to shape the future of any rebate-based system by submitting comments on:
- Potential risks to cash flow or patient access.
- IT, staffing, or workflow burdens.
- The adequacy of safeguards currently proposed.
- Suggestions to improve transparency or operational ease.
How to Submit Feedback
Visit www.regulations.gov and search for the document HRSA-2025-0001. All comments are due by September 8, 2025.
340B Proves Critical in Emergencies and Everyday Care
As policy debates continue around the future of the 340B Drug Pricing Program, new developments in 2025 highlight both the program’s immediate impact during crises and its long-term value in underserved communities.
In June, the Health Resources and Services Administration (HRSA) took a major step by allowing providers in disaster-designated areas to bypass normal enrollment windows and access 340B pricing immediately. This emergency policy was activated following severe flooding in parts of Georgia and Texas, giving affected health centers and hospitals the ability to secure affordable medications within days rather than waiting for the next quarterly registration period.
“HRSA recognizes the critical need to maintain affordable access to life-saving medications during emergencies,” the agency stated. Providers in five Texas counties were among the first to benefit, helping patients receive medications like insulin, inhalers, and antibiotics without delay, despite pharmacy closures and infrastructure disruptions.
Policy Advocacy Extends Beyond Emergencies
At the same time, Texas healthcare leaders are speaking out about the importance of preserving 340B access across the board. In a June 7 San Antonio Express-News op-ed, hospital administrator Gina Ramirez and pediatrician Dr. Samir Khoury stressed that 340B is the financial backbone of services that many communities depend on—not only during disasters, but every day.
“For many of our patients, these are not just lower prices. These are the only prices they can afford,” they wrote. They emphasized how 340B savings help clinics offer behavioral health, substance use treatment, and in-house pharmacy services that would otherwise be unaffordable or unavailable—particularly in rural towns and border communities.
The op-ed supports Texas House Bill 3265, bipartisan legislation that would protect 340B access at the state level. Advocates say the bill is vital to ensuring the program’s future in regions where patients face high costs, long travel distances, and limited healthcare infrastructure.
Together, these developments paint a clear picture: whether it’s responding to a public health emergency or sustaining primary care in under-resourced areas, the 340B program is more than a discount – it’s a lifeline. As Ramirez and Khoury concluded, “When you protect 340B, you protect patients.”
New Executive Order Expands Access to Affordable Insulin and EpiPens
A recent Executive Order requires Federally Qualified Health Centers (FQHCs) receiving Section 330 grant funding to provide low-cost insulin and injectable epinephrine to low-income patients through the 340B drug pricing program.
Scope of the Policy
This policy applies only to patients served by these federally funded health centers and excludes Look-Alike centers that do not receive Section 330 funding. While there is no formal start date, health centers are expected to update their policies promptly and be prepared to show compliance during HRSA site visits.
Eligibility, Medications, and Pricing Requirements
Discounted pricing must be available through in-house pharmacies, contract pharmacies, or provider-dispensed models. Covered medications include all insulin products and injectable epinephrine, such as EpiPen. It does not apply to GLP-1 medications like Ozempic or to non-injectable epinephrine.
Eligibility is limited to patients whose household income is at or below 200 percent of the Federal Poverty Guidelines. Health centers should prioritize individuals with high out-of-pocket costs, large unmet deductibles, or no health insurance. Many drug manufacturers restrict access to 340B-priced insulin at contract pharmacies. Health centers may designate one contract pharmacy per site and must document their efforts to comply. If medications are unavailable at 340B prices, discounts cannot be offered.
Policy Updates, Patient Communication, and Compliance Resources
Though not required, informing patients about where to access these medications at reduced costs remains a recommended best practice. There is no federal definition for a “minimal administration fee,” so centers may refer to state Medicaid dispensing rates or use tools like the Apexus 340B Medicaid Profiles to guide their pricing.
Health centers can either revise their current Sliding Fee Scale policy or create a new one to meet this requirement. Policies should define terms such as high cost-sharing, high unmet deductible, and lack of insurance, aligned with the center’s income guidelines.
More information is available from the 340B Prime Vendor Program (PVP) and the National Association of Community Health Centers.
What to Expect Next
FQHCs must update policies now to offer 340B-priced insulin and injectable epinephrine to eligible low-income patients. While no start date is set, HRSA will check compliance during site visits. Allowable access points include in-house, provider-dispensed, or one contract pharmacy per site. Clear documentation and patient communication are encouraged. Readiness is expected promptly.
Capitol Hill Sees Record Attendance at ‘Defend 340B’ March
On July 24, nearly 100 advocates gathered for the fourth annual ‘Defend 340B’ March in Washington, D.C., marking the event’s largest turnout to date. Taking place just after the 340B Coalition Conference, the march aimed to raise awareness of the program’s role in advancing health equity across underserved communities. As challenges to 340B continue to mount, advocates are stepping up louder and stronger, demonstrating the program’s critical value to providers and patients nationwide.
“What 340B does is ensure that we can take care of people in our communities that need help the most.” – John Carlo, CEO, Prism Health North Texas
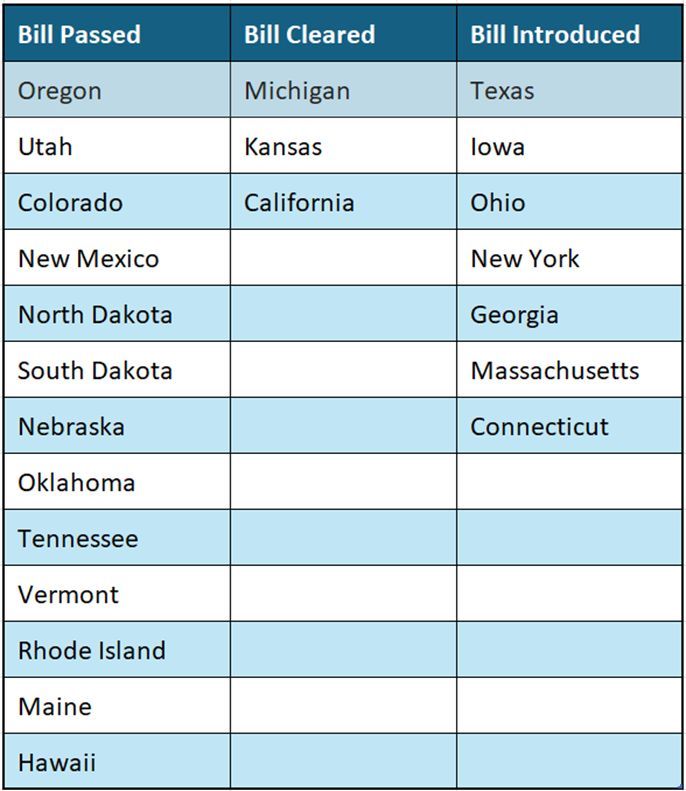
Current Status on State Bills and Laws That Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates since last month
Appeals Court Revives 340B Antitrust Lawsuit Against Major Insulin Manufacturers
On August 6, 2025, the U.S. Court of Appeals for the Second Circuit unanimously reinstated an antitrust lawsuit brought by Mosaic Health (NY) and Central Virginia Health Services against AstraZeneca, Eli Lilly, Novo Nordisk, and Sanofi. The ruling reverses a 2024 district court dismissal, allowing the case to proceed in the Western District of New York.
The suit alleges the manufacturers unlawfully coordinated policies restricting access to 340B discounts through contract pharmacies, beginning in 2020. The appeals court found enough evidence—similar timing, substance, and effect of the restrictions, along with other “plus factors”—to plausibly suggest a horizontal price-fixing conspiracy in violation of the Sherman Antitrust Act. The case does not challenge the legality of contract pharmacy restrictions under the 340B statute, but rather whether they resulted from collusion.
Rare Antitrust Approach in 340B Disputes
The decision is notable because antitrust claims involving the 340B program are rare. By framing the dispute around competition law rather than program compliance, the plaintiffs have opened a new legal front that could resonate beyond the insulin market. Advocates say it highlights a pathway for covered entities to challenge manufacturer practices that erode program value.
With the ruling, the plaintiffs can file a second amended complaint and move into discovery, which is expected to probe internal communications, policy development, and industry coordination. For covered entities, this decision marks a rare opportunity to test whether antitrust law can be used to push back on manufacturer practices that limit program savings and patient care capacity.
What to Expect Next
The case’s progress will be closely watched by 340B stakeholders. A win for the plaintiffs could reshape the legal landscape, deter coordinated manufacturer restrictions, and prompt greater transparency in policy decisions affecting contract pharmacy access.

