Key Updates at a Glance:
- Expanded Manufacturer Reporting: Eli Lilly and Novo Nordisk are now conducting in-house claims reporting, increasing operational and compliance responsibilities for covered entities.
- Contract Pharmacy and ADR Trends: Recent ADR decisions continue to favor manufacturers, reinforcing limitations around contract pharmacy arrangements.
- HRSA Rebate Model Activity: HRSA has extended the RFI deadline to April 20; bipartisan lawmakers are actively pushing to block the proposed rebate model.
- Federal Litigation Involvement: DOJ and HHS are supporting manufacturers in state-level 340B cases, adding legal complexity.
- State-Level Developments: Minnesota’s recent ruling upheld contract pharmacy access, indicating continued state-level support for the program.
- Policy Pressure Building: Both federal agencies and Congress are signaling increased scrutiny and potential intervention on contract pharmacy and rebate issues.
340B In-House Pharmacy Data Reporting Sparks Pushback
Eli Lilly and Novo Nordisk have expanded 340B claims reporting to include all in-house
pharmacy activity. Lilly requires providers to submit claims data within 45 days, while Novo
Nordisk will require pharmacy and medical claims data for every 340B dispense starting April 1.
Previously, reporting focused mostly on contract pharmacies. Manufacturers say these policies
increase transparency and protect program integrity. Providers argue they create significant
operational and financial strain, particularly for under-resourced hospitals and clinics.
Operational and Financial Implications
Covered entities may need new electronic medical record integrations, additional staffing, or
vendor support to handle the increased data volume. AEH (America’s Essential Hospitals)
reported essential hospitals averaged a -7.1% operating margin in 2023, highlighting limited
capacity to absorb these costs.
For CEs, this translates into:
- Diverted staff time from patient care to data reporting.
- Budget pressure from IT upgrades or compliance support.
- Potential workflow disruptions in pharmacy operations.
Smaller clinics and federally qualified health centers are particularly vulnerable, as they already
manage substantial reporting obligations.
Compliance and Legal Considerations
Manufacturers cite HRSA guidance permitting reasonable information requests from CEs. Experts
say reporting requirements can be lawful, but the scale of these new obligations may push the
limits of what’s reasonable under the 340B statute. Failure to submit complete or timely data
could result in suspension of 340B pricing, adding financial risk and operational uncertainty.
Many CEs are reviewing compliance processes and considering third-party support to mitigate
disruptions.
Implications for Covered Entities
While manufacturers present these policies as strengthening program oversight, the practical
effect is increased administrative and financial pressure on hospitals and clinics. CEs may need to
reconsider:
- Staffing and resource allocation.
- In-house pharmacy scope and operations.
- Contract pharmacy strategies to balance reporting demands with patient care priorities.
What to Expect Next
Covered entities should assess reporting capabilities immediately. HRSA has not formally
weighed in on the legality of these requirements, and any future agency guidance could shift
manufacturer‑CE interactions. Other drugmakers may follow, potentially making comprehensive
in-house pharmacy reporting the new standard. Provider advocacy will continue, but CEs must
act now to avoid disruptions and protect patient access.
340B Contract Pharmacy Limits Reinforced by Latest ADR Decisions
A 340B Administrative Dispute Resolution (ADR) panel recently rejected a hospital challenge to a A340B Administrative Dispute Resolution (ADR) panel recently rejected a hospital challenge to a drugmaker’s 340B contract pharmacy restrictions, marking the sixth consecutive time an ADR panel has sided with manufacturers under the updated process.
The latest ruling dismissed a petition from Wisconsin-based St. Croix Regional Medical Center against Danish drugmaker Novo Nordisk’s contract pharmacy limitations, citing a January 2023 Philadelphia federal appeals court decision that upheld similar restrictions. The panel found “no overcharge violation” under the 340B statute.
The decision does not address Novo Nordisk’s newly announced requirement for covered entities (CEs) to submit claims data for in-house pharmacy dispenses to access 340B pricing, a policy set to take effect April 1 that providers argued is unlawful and that the company says enhances transparency.
Background on the ADR Process
The ADR process was established under the Affordable Care Act in 2010 as the primary federal mechanism for resolving disputes over 340B pricing and alleged overcharges. HRSA did not finalize its first ADR rule until 2020, later withdrew it for reworking, and released a new final rule that took effect in June 2024.
Under the current framework, a three-member panel from HRSA’s Office of Pharmacy Affairs reviews submitted claims and issues final decisions, which are publicly summarized within 120 days. Some providers have criticized HRSA for delays in reviewing pending claims. In September 2025, multiple health systems sued the agency over an “unreasonable delay” in processing an ADR petition against Teva Pharmaceuticals.
What This Means for Covered Entities
For hospitals and other CEs, the latest ruling signals that challenging manufacturer-imposed limits through ADR will remain difficult:
- Few Legal Options: With all recent ADR rulings favoring manufacturers, CEs may find limited resources to contest contract pharmacy restrictions.
- Operational Adjustments: Facilities may need to re-evaluate the number of contract pharmacies they use or shift more activity to in-house pharmacies to maintain compliance and preserve 340B savings.
- Financial Considerations: Stricter limits could affect revenue derived from contract pharmacies, requiring careful planning to manage potential impacts on patient services.
Looking Ahead
The repeated ADR rulings underscore that manufacturers’ contract pharmacy policies are likely to remain enforceable under current federal guidance. While separate antitrust or state-level litigation could eventually provide alternate pathways for challenges, CEs should prepare for ongoing limitations on contract pharmacy operations. Proactively, covered entities may need to review and optimize their contract pharmacy networks, strengthen compliance documentation, and ensure operational flexibility to maintain access to 340B pricing under these policies.
Expanding In-House Reporting and HRSA RFI Update
HRSA continues exploring updates to the 340B program, including potential rebate models for Part D and IRA drugs. While implementation is paused, recent manufacturer policies highlight growing overlap between federal and private reporting requirements for covered entities.
Data Requirements and Operational Overlap
Both Eli Lilly and Novo Nordisk recently implemented policies requiring CEs to submit pharmacy and medical claims data for in-house 340B dispensing. While framed as transparency measures, these policies create operational and compliance overlap with data HRSA might seek if it moves forward with a rebate model.
CEs may face situations where:
- The same claims and acquisition data are used to satisfy both manufacturer requirements and potential HRSA reporting standards.
- Data collected for manufacturers could later be referenced to justify a federal rebate approach or validate pricing compliance.
- Operational systems must accommodate additional recordkeeping, cross-checking, and IT governance to ensure accuracy across multiple reporting obligations.
These parallel demands raise strategic questions: how to manage overlapping submissions, reconcile claims, and anticipate operational or financial exposure if HRSA leverages the information for a future rebate model.
HRSA RFI Update
HRSA’s Request for Information (RFI), first published in February, seeks stakeholder feedback on how a 340B rebate system could be structured if pursued. The agency is requesting input from hospitals, manufacturers, pharmacies, wholesalers, and state Medicaid agencies on:
- Administrative and cash-flow impacts of shifting from upfront discounts to rebates
- Safeguards to prevent duplicate discounts
- Data and transparency improvements for program oversight
HRSA recently extended the comment period to April 20, 2026, giving stakeholders additional time to provide input. While the RFI does not impose immediate requirements, submissions could shape HRSA’s thinking on the scope, data needs, and operational design of any future rebate model.
Covered entities should ensure they can:
- Accurately report claims data
- Streamline IT and compliance processes
- Consider how submissions might influence HRSA policy or future 340B program changes
Looking Ahead
The RFI remains a neutral information-gathering step, but it signals continued evaluation of a federal rebate model. For CEs, the key focus is on understanding the overlap between current manufacturer data requests and possible federal requirements. Proactive preparation now can help mitigate operational strain and ensure data integrity, regardless of whether a rebate model is ultimately implemented.
To view HRSA’s RFI docket, click here.
Federal Government Joins State 340B Contract Pharmacy Disputes
The federal government has now entered state 340B contract pharmacy litigation, siding with drugmakers in amicus briefs filed in Colorado and Rhode Island. The U.S. Departments of Justice (DOJ) and Health and Human Services (HHS) urged federal appellate courts to overturn state laws expanding contract pharmacy access, arguing these statutes conflict with the federal 340B program.
The briefs were signed by DOJ Assistant Attorney General Brett Shumate and attorney Yaakov Roth, both of whom previously represented drugmakers in 340B-related litigation. Provider attorneys have questioned how much weight the government’s arguments will carry, given prior federal court rulings favoring state 340B access laws.
State Litigation Developments
- Louisiana: The Louisiana Primary Care Association asked the 5th Circuit to reconsider a procedural ruling on its intervention in a case upholding the state’s contract pharmacy law.
- Rhode Island: PhRMA filed an opening brief asking the 1st Circuit to overturn a lower court ruling upholding the state’s 2025 law, which prevents manufacturers from conditioning 340B access on claims data. The federal government filed an amicus brief backing PhRMA.
- Oklahoma: The state attorney general filed briefs in two 10th Circuit appeals defending Oklahoma’s law, which remains enjoined following preliminary rulings in favor of drugmakers.
- Colorado: AbbVie appealed a lower court ruling denying a preliminary injunction against the state’s 2025 law, with the federal government supporting the manufacturer.
- Minnesota: A state appeals court upheld Minnesota’s contract pharmacy law, creating tension with a prior federal ruling that found the statute unenforceable.
Manufacturer Updates
Amgen recently revised its contract pharmacy policy, exempting Oregon providers while removing a prior exemption for New Mexico. Its policy generally limits 340B shipments to an in-house or single contract pharmacy when claims data are submitted, highlighting ongoing operational complexity for covered entities navigating varying state and manufacturer requirements.
Implications for Covered Entities
- CEs should monitor federal and state litigation closely, as decisions in any of these cases could set precedent affecting multiple states.
- Contract pharmacy agreements may need periodic review to ensure compliance with both state laws and manufacturer restrictions.
- Operational systems must be flexible to manage reporting, claims reconciliation, and inventory across differing manufacturer requirements and evolving legal landscapes.
Looking Ahead
The interplay between federal advocacy for drugmakers, ongoing state court victories for CEs, and manufacturer policy adjustments signals a period of continued uncertainty. Covered entities should proactively align legal, compliance, and pharmacy operations to navigate both current rules and potential rapid shifts in contract pharmacy access and reporting obligations.
Minnesota Decision Offers Encouraging Outlook for 340B Providers
A recent Minnesota state appeals court decision upheld the state’s 2024 contract pharmacy law, confirming that manufacturers cannot restrict 340B drug delivery to contract pharmacies under state law. This ruling affects more than 200 covered entities in Minnesota, including hospitals and community health centers, and safeguards access for roughly 350,000 patients relying on 340B discounts.
This decision is a clear win for providers within the state, reinforcing the ability of states to protect 340B program access even amid federal and manufacturer pressures. Minnesota now joins over 15 states that have enacted laws to preserve contract pharmacy access, providing stronger operational certainty for providers.
By maintaining contract pharmacy access, providers within Minnesota can continue serving vulnerable populations without disruption. The ruling also offers clearer guidance for managing 340B operations, including inventory, reporting, and pharmacy partnerships, helping maintain program integrity while supporting patient care.
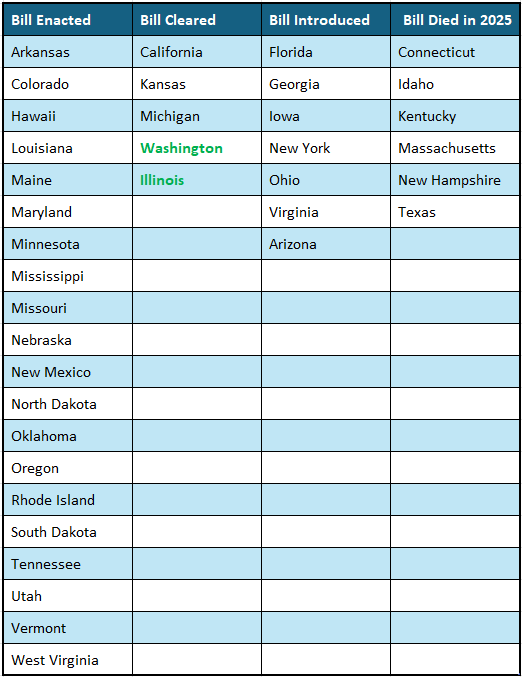
Current Status on State Bills and Laws That Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates.
Washington Update: 340B Remains in the Spotlight
Since the Feb. 11 House Energy & Commerce hearing, 340B has continued to draw attention on both the federal and state levels, with new proposals, bipartisan advocacy, and court actions fueling ongoing scrutiny and potential operational impacts for covered entities (CEs).
Bipartisan Push Against HRSA Rebate Model
A group of key bipartisan lawmakers is pushing to include language in a must-pass federal appropriations bill that would block implementation of a 340B rebate model. U.S. Reps. Doris Matsui (D-Calif.), Dusty Johnson (R-S.D.), Debbie Dingell (D-Mich.), and Tracey Mann (R-Kan.) circulated a letter urging Congress to prevent HRSA from requiring retroactive rebates, citing concerns over administrative burden and cash flow challenges for safety-net providers. The letter received at least 40 signatures as of March 17, 2026.
Federal Appeals Court Reinstates 340B Overcharge Lawsuit
On March 17, the U.S. Court of Appeals for the 9th Circuit revived a whistleblower lawsuit under the False Claims Act involving four major drugmakers — AbbVie, AstraZeneca, Novartis, and Sanofi — alleging they overcharged government programs in connection with 340B drug pricing. The case will return to district court, marking an important development in legal oversight of manufacturer compliance with 340B discount requirements.
Why This Matters to Covered Entities
These developments reinforce that 340B remains an active focus of policy and legal scrutiny. The reinstated False Claims Act litigation could shape how manufacturer compliance is enforced, potentially increasing oversight expectations. At the same time, bipartisan efforts to block a rebate model signal continued debate, but also support, for maintaining the program’s current structure.
Covered entities should:
- Monitor state Medicaid policy proposals in states considering reimbursement changes tied to 340B purchases.
- Stay apprised of ongoing litigation, especially False Claims Act cases affecting manufacturer compliance.
- Engage with policymakers early, using data to illustrate how 340B savings support patient access and safety-net services.
As 2026 progresses, legal activity, state policy changes, and broader drug pricing debates will continue shaping the 340B program, making vigilance and proactive planning essential for covered entities.

