This Month at a Glance
- Contract pharmacy litigation turns against providers. First appellate losses in the 4th Circuit and a North Dakota ruling striking down state law; Washington adds new lawsuits, while Louisiana signals active enforcement against manufacturers.
- 340B rebate model remains unresolved. HRSA received 5,500+ comments but has not acted; Congress remains split, leaving timing and scope uncertain.
- In-house 340B data requirements expand. Eight manufacturers extend claims-level reporting to in-house pharmacies; HRSA has not taken a position.
- Indiana eliminates most Medicaid 340B savings. State replaces spread-based reimbursement with rebates, creating a potential model for other states.
- Tennessee tightens eligibility rules for in-kind funded clinics. New audit-based requirements for 340B participation amid rising manufacturer scrutiny of clinic eligibility.
- Child site ruling under appeal. HRSA appeal puts hospital outpatient access in legal uncertainty while current access remains in place.
State Contract Pharmacy Laws Face New Legal Headwinds
After a sustained run of provider wins in court, the legal landscape shifted. Drugmakers notched their first-ever appellate victories against state contract pharmacy access laws, a federal judge in North Dakota struck down that state’s law in unusually harsh terms, and the Trump administration’s Justice Department escalated its involvement on the manufacturer side, intervening at the district court level for the first time. The picture varies by state, but CEs across the country are operating with more uncertainty than they were three months ago.
First Appellate Losses for Providers
The U.S. 4th Circuit Court of Appeals issued 2-1 rulings in April striking down contract pharmacy access laws in both West Virginia and Maryland, the first times a federal appeals court has ruled against a state contract pharmacy law. The same two Trump-appointed judges wrote both decisions using the same legal theory; a Biden-appointed judge dissented in both. Both states have asked the full 4th Circuit to reconsider, backed by amicus briefs from AHA, 340B Health, and state hospital associations. Full-court rehearing is rarely granted, but the court has a slightly Democratic-leaning composition, and the stakes are high enough that it cannot be ruled out.
A federal judge in North Dakota struck down that state’s law on April 27 in a ruling notable for its sharp rhetoric, accusing providers and pharmacies of exploiting the program. The state appealed immediately and has reason for optimism. The 8th Circuit unanimously upheld Arkansas’ similar law in 2024, and the state’s opening brief is due June 15. Utah, meanwhile, offers a more encouraging model: after a 2025 ruling flagged a definitional problem in its law, the legislature amended the statute, and the state attorney general has since moved to dismiss all three pending manufacturer lawsuits.
Washington and Louisiana
Washington’s new contract pharmacy access law, the 22nd in the nation, takes effect June 10 and has already drawn four manufacturer lawsuits from AbbVie, Novartis, PhRMA, and AstraZeneca. Louisiana is moving in the other direction: the state attorney general announced she is preparing enforcement actions against manufacturers violating the state’s contract pharmacy law, making it the first state to signal active enforcement rather than waiting for courts to act.
What This Means for CEs
CEs in Maryland and West Virginia should seek legal guidance on handling manufacturer restrictions while their state’s law remains blocked. In states where laws have been upheld (Louisiana, Mississippi, and Arkansas) those protections remain in force, though the federal government is actively working to reverse the Louisiana ruling.
Washington state CEs should monitor litigation closely ahead of the June 10 effective date. Regardless of where you operate, do not assume your state’s law will hold, this legal landscape is moving faster than at any point since these disputes began.
340B Rebate Model: Still No Decision Despite Record Number of Comments
HRSA’s request for information on a potential 340B rebate model closed April 20 after more than 5,500 submissions, over four times the volume received on last year’s halted pilot. Despite this response, the Trump administration has not indicated next steps. Uncertainty grew the day after the deadline, when HHS Secretary Robert F. Kennedy Jr. told a Senate panel he did not know where the proposal currently stood.
Stakeholders Remain Deeply Divided
Providers argued the model would create major operational and financial strain:
- The American Hospital Association estimated hospitals could face more than $1 billion annually in added costs and signaled potential litigation if HRSA moves forward.
- Community health centers warned that compliance costs alone could exceed $3 million annually for a single mid-sized organization.
- Many covered entities emphasized the cash flow burden of purchasing drugs at full cost while waiting for reimbursement.
Manufacturers pushed aggressively in the opposite direction:
- PhRMA urged HRSA to implement a rebate model by January 1, 2027.
- Manufacturers argued any future model should apply broadly across 340B purchases rather than operate as a narrow pilot.
- Johnson & Johnson stated it has already incurred $80 million in duplicate discounts this year tied to Medicare negotiated drugs, using that figure to argue for urgent policy action.
Where Things Stand
HRSA has not issued a formal proposal, but a February filing suggests the agency is still considering a 2027 pilot limited to Medicare-negotiated drugs, essentially the prior framework, delayed one year.
Congress is divided. More than 100 bipartisan lawmakers have urged appropriators to block any rebate model in FY2027 spending legislation, while seven House Republicans have encouraged the administration to proceed.
Lawmakers are also considering narrower protections. A bipartisan bill from Reps. Bergman and Ruiz would exempt health centers, critical access hospitals, and Ryan White clinics from any future rebate model, though disproportionate share hospitals and other large systems would not be included.
What This Means for CEs
A rebate model has not been announced, but risk and timing remain uncertain. Covered entities should assess cash flow exposure from purchasing at full cost while awaiting reimbursement, especially for Medicare-negotiated drugs, which remain the most likely initial scope.
Health centers and rural providers should monitor the Bergman-Ruiz bill, as it could provide a targeted safe harbor. More broadly, the FY2027 appropriations process will be key, rebate-blocking language could offer the strongest near-term protection. While HRSA has not formally advanced a rebate model, the volume of stakeholder engagement and continued manufacturer pressure indicate the issue remains very active. Covered entities should continue monitoring both regulatory and legislative developments closely, particularly as FY2027 appropriations negotiations and manufacturer litigation strategies evolve.
HRSA Silent as In-House Pharmacy Requirements Expand
What started as a contract pharmacy compliance issue has evolved into a broader challenge for covered entities’ in-house dispensing operations. Six manufacturers now require claims-level data for drugs dispensed through in-house pharmacies as a condition of continued access to 340B pricing across both contract and owned facilities. Biogen announced on May 1 that it will require in-house claims data submissions for four multiple sclerosis drugs beginning June 1. More recently, Amgen announced on May 15 that it will also implement medical claims submission requirements effective June 1. Despite months of requests from provider groups, HRSA has not stated whether these policies are lawful.
A Growing List of Manufacturers
Eight manufacturers now have active or upcoming in-house pharmacy data requirements: Exelixis, Eli Lilly, Novo Nordisk, AstraZeneca, Bristol Myers Squibb, Biogen, Amgen, and UCB. Together, these policies span oncology, diabetes, hepatitis C, cardiovascular disease, and multiple sclerosis. Most require covered entities to submit claims-level data within 45 days to maintain 340B pricing on affected drugs, with noncompliance potentially resulting in loss of access.
State exemptions vary significantly:
- Biogen exempts 13 states
- Lilly exempts 11 states
- Bristol Myers Squibb exempts 4 states
- Novo Nordisk and AstraZeneca apply their policies nationwide with no exemptions
Covered entities should not assume exemptions apply consistently across manufacturers.
A growing disconnect is also emerging between manufacturers’ publicly posted policy language and the operational requirements appearing within 340B ESP workflows. Several manufacturers appearing on 340B ESP operational NDC lists requiring medical claims data submissions, including AbbVie, Boehringer Ingelheim, Johnson & Johnson, Merck, and Novartis, continue using narrower public-facing language focused on “limited claims data,” contract pharmacy claims, or specialty pharmacy controls rather than explicitly referencing medical claims or in-house dispensing data requirements.
This has created uncertainty around whether ESP operational expectations extend beyond what manufacturer policy notices formally describe.
Escalating Operational Pressure
The issue has now moved from policy debate to active enforcement pressure. Covered entities report receiving warning letters from Eli Lilly and Company indicating potential loss of 340B pricing for hospitals that have not complied with data submission requirements.
At the same time, the American Hospital Association has sent a third letter to HRSA raising concerns that manufacturers are effectively redefining program requirements without federal clarification. HRSA has not issued a public position, while providers and manufacturers continue to disagree on whether these requirements exceed the scope of the 340B statute.
What This Means for CEs
Covered entities should immediately assess exposure across all six manufacturers and identify:
- Which drugs are affected under in-house dispensing policies
- Whether any state-level exemptions apply
- Whether current systems can meet 45-day reporting requirements
Entities should not wait for HRSA guidance, as no timeline has been provided for intervention. For hospitals receiving Eli Lilly warning letters, legal review is strongly advised before responding or complying, as both compliance and noncompliance carry financial risk depending on downstream enforcement outcomes.
Indiana Moves to End 340B Savings Under Medicaid
Indiana will become one of the most significant tests of state Medicaid 340B policy beginning July 1, when a new rule eliminates most covered entities’ ability to generate 340B savings under Medicaid managed care. The change stems from a provision inserted into the state’s May 2025 budget law, added the night before final passage without public hearings, granting broad authority over 340B Medicaid reimbursement. After provider advocacy, federally qualified health centers received an exemption; hospitals did not.
What Changes on July 1
Currently, Indiana covered entities purchase drugs at the 340B acquisition price and bill Medicaid managed care plans at standard rates, retaining the spread. Under the new policy, the state will instead collect manufacturer rebates directly, effectively eliminating 340B savings for all covered entities except FQHCs.
Indiana estimates the policy will save Medicaid roughly $63 million annually, though hospitals note the state retains only about one-third of that amount, with the remainder flowing to the federal government. The Indiana Hospital Association argues the change reduces safety-net funding more than it benefits the state budget and could shift costs to commercial payers and reduce access to care.
The FQHC exemption followed sustained advocacy from the Indiana Primary Health Care Association and was framed by state officials as consistent with Governor Mike Braun’s focus on protecting low-income patients. Hospitals, while appreciative of the carveout, continue to oppose the broader policy. The July 1 effective date remains unchanged.
Broader Policy Signal
Indiana’s approach reflects a broader shift in how states may begin targeting 340B savings through Medicaid payment redesign rather than direct program restrictions. Unlike traditional contract pharmacy or eligibility disputes, this model intervenes at the reimbursement structure level, making it potentially more durable and harder to challenge under existing 340B enforcement pathways.
A Potential Model for Other States
Indiana is not the first state to alter Medicaid 340B reimbursement. California and New York have done so previously, but the way this change happened matters. It came through a budget process rather than standalone legislation, was added at the last minute without public debate, and took effect without any formal rulemaking. That path is available to any state legislature, and as Medicaid budgets come under pressure and awareness of 340B savings grows among state officials, Indiana’s approach may attract attention elsewhere.
What This Means for CEs
Indiana FQHCs should confirm the exemption is formally reflected in the state plan and effective July 1. Indiana hospitals should immediately model financial impacts, including which managed care claims currently generate 340B savings and what programs those revenues support.
For covered entities outside Indiana, the key takeaway is procedural as much as policy: this type of change can occur quickly through budget legislation with limited visibility. It is worth elevating this model with state hospital and health center associations as a potential emerging risk pathway.
Tennessee Strengthens Eligibility Requirements for In-Kind Funded Clinics
Tennessee Gov. Bill Lee signed legislation on April 27, tightening eligibility requirements for nonprofits receiving in-kind funding, such as immunization and STD clinics, to qualify as 340B covered entities. Starting November 1, these organizations must also receive at least one state or federal grant subject to formal audit and oversight to maintain eligibility. The law builds on a 2024 statute that added contracting requirements for similar clinics.
Amgen and Novartis have recently ended 340B pricing for STD clinics, arguing their products are not indicated for STD treatment, and manufacturers filed suit in December 2024 claiming ineligible clinics improperly accessed 340B savings. Tennessee’s law responds to this scrutiny by strengthening compliance standards for in-kind grantees.
STD and TB clinics operating under 340B should be aware that manufacturer eligibility challenges are increasing nationwide. States without clear rules for in-kind grantees may leave clinics more exposed, making compliance reviews ahead of the November 1 deadline important.
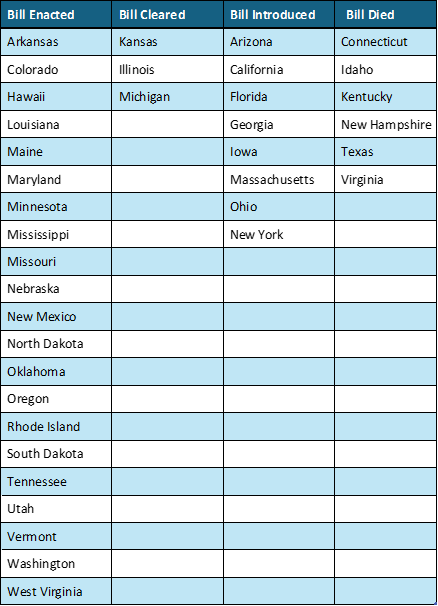
Current Status on State Bills & Laws That Prohibit 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates.
HRSA Appeal Reignites Child Site Dispute
A significant hospital win earlier this year is now headed to federal appeals court. HRSA filed a notice of appeal in late April challenging the March 3 district court ruling that struck down the agency’s longstanding requirement that hospitals complete formal registration before accessing 340B pricing at new offsite outpatient facilities (child sites). The ruling was an important victory for hospitals, which had argued the process can take years and delay access to eligible sites. With HRSA now appealing, that win is no longer settled law.
The History of the Policy
HRSA requires new hospital outpatient facilities to be listed on the Medicare cost report and formally registered before accessing 340B pricing. The agency waived this requirement during the COVID-19 public health emergency and later stated the waiver was not tied to the emergency itself. When the PHE ended in May 2023, HRSA reinstated the policy, prompting hospital litigation.
In March, Judge Amit Mehta ruled that the requirement “conflicts with the text of the 340B statute,” noting that HRSA’s prior statements weakened its authority to enforce it. HRSA’s appeal signals it still views the policy as central to program oversight.
What Hospitals Should Know Now
The ruling remains in effect during appeal, meaning hospitals may currently access 340B pricing at eligible child sites pending registration. However, HRSA could seek a stay, which would immediately reinstate the registration requirement.
Hospitals that have already accessed 340B pricing at unregistered sites should be aware that a stay or reversal could create compliance exposure. The D.C. Circuit has set initial briefs due June 1, with a final decision unlikely before late 2026 at the earliest.
What This Means for CEs
This decision should not be treated as final. Hospitals that have relied on the ruling to access 340B pricing at new child sites should consult counsel regarding potential exposure if the policy is reinstated on appeal.
At the same time, providers should continue completing HRSA’s formal registration process where possible to maintain compliance under either outcome. For hospitals planning outpatient expansion, the key takeaway is uncertainty: 340B eligibility at new sites is not guaranteed while the appeal is pending.

