HRSA’s Rebate Pilot, Contract Pharmacy Legal Battles, PHRMA’s New Campaign, Eliquis WAC Reduction, and more in this month’s edition of ACI Monthly.
HRSA’s 340B Rebate Pilot to Launch January 1, 2026
HRSA has approved eight manufacturer rebate plans covering nine drugs under the new 340B Rebate Model Pilot, beginning January 1, 2026.
Approved Manufacturers and Drugs
- Bristol Myers Squibb – Eliquis
- Amgen – Enbrel
- AstraZeneca – Farxiga
- Pharmacyclics – Imbruvica
- Merck – Januvia
- Novo Nordisk – Fiasp, Novolog
- Boehringer Ingelheim – Jardiance
- Johnson & Johnson – Stelara, Xarelto
- Novartis – Entresto (beginning April 1st, 2026)
Novartis’ Entresto was not included in the initial wave of approvals. Due to this, the start date has been pushed to April 1st, 2026. Novartis, like other manufacturers, must provide covered entities with 60 days’ notice before the start date.
For more detailed information on each drug approved, you can download “Beacon’s NDC List” at the bottom of their resource page by clicking here.
Ordering and Submission Timing
Covered entities may continue ordering as usual leading into the new year. Some manufacturers may permit December claims submissions to account for partial dispensing, but this will vary by plan.
No rebate submissions can be made until January 2, 2026, and only after a qualifying WAC purchase has occurred in the 340B account.
Updated Data Requirements
HRSA’s October 30 approval introduced a defined set of medical-claims fields that must accompany standard pharmacy data for rebate submissions, and Beacon’s website now outlines these requirements. Fields such as the NDC, quantity, physician NPI, service-provider NPI, and date of service are already common in claim records and should be straightforward for contract-pharmacy workflows.
The greater impact will be in split-bill settings, where TPAs may need additional information from patient files. The most challenging elements are expected to be the claim number and the health plan ID, as these identifiers are not always included in the patient-level files currently provided by internal IT teams. Beacon’s final technical specifications remain on track for release in December 2025. Make sure to review Beacon’s welcome package and review the Pharmacy Claims and Medical Claims section.
Preparing for January 1
- Register in Beacon promptly.
- Coordinate pharmacy, compliance, and IT teams around the 45-day claim window.
- Model cashflow impact as rebates replace up-front discounts.
- Monitor HRSA, Beacon, and Apexus for additional guidance.
How ACI Can Help
ACI is assisting covered entities through this transition by supporting Beacon registration and setup, performing impact analyses to project financial effects, and assisting in managing claims-data submission once the program begins. Most TPAs are still developing the technical processes needed to support this rebate workflow.
A Growing Legal Battle Over Contract Pharmacies
The legal landscape surrounding 340B contract pharmacy access continues to expand, with manufacturers intensifying efforts to challenge state laws designed to preserve access to discounted drugs for covered entities. These laws, which require drugmakers to honor 340B pricing for contract pharmacies, have become a central point of contention between states and the pharmaceutical industry.
AstraZeneca Challenges Vermont’s Contract Pharmacy Law
On November 4, AstraZeneca filed a new lawsuit in the U.S. District Court for the District of Vermont, marking the second legal challenge to the state’s 340B contract pharmacy access law. The company argued that the statute conflicts with the federal 340B program and patent law, unlawfully interferes with private contracts, and constitutes an unconstitutional taking. AstraZeneca also claimed that Vermont’s law would prevent it from participating in HRSA’s 340B rebate pilot program, which launches January 1, 2026, because the law prohibits manufacturers from collecting certain claims data.
Vermont’s law, effective June 11, 2025, bars manufacturers from restricting the delivery of 340B drugs to contract pharmacies or conditioning discounts on data reporting not required under federal law. It also imposes penalties of up to $100 per package for violations.
Expanding Litigation Nationwide
AstraZeneca’s complaint adds to a growing wave of more than 50 ongoing lawsuits across the country challenging state contract pharmacy access laws. Drugmakers including AbbVie and industry group PhRMA have mounted similar challenges in states such as Oregon, Nebraska, Hawaii, Mississippi, Arkansas, Oklahoma, and West Virginia.
To date, most federal district courts have sided with states: judges in nine states have upheld contract pharmacy laws, and the 5th and 8th Circuit Courts of Appeals have affirmed rulings favoring Mississippi and Arkansas. However, preliminary injunctions in Oklahoma and West Virginia have temporarily halted enforcement there.
Why It Matters for Covered Entities
For covered entities, these lawsuits create continued uncertainty around how many contract pharmacies can participate, what data manufacturers may require, and whether certain drugmakers may restrict pricing while litigation is pending. Entities reliant on external pharmacies, especially rural hospitals and clinics, could face operational or financial strain if manufacturers limit participation or delay shipments.
Preparing for Potential Impact
- Maintain up-to-date documentation for all contract-pharmacy relationships to enable rapid compliance reviews and risk assessments if state laws or manufacturer policies change.
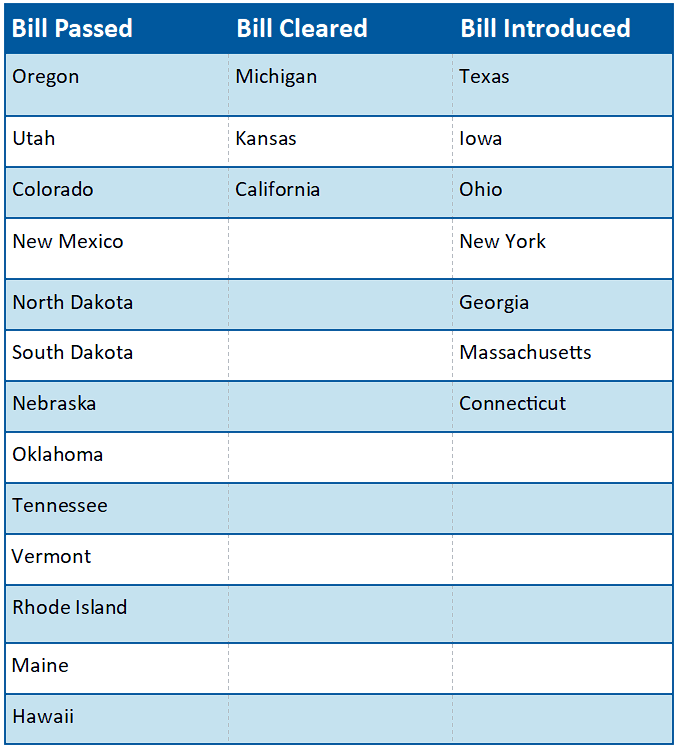
- Track legislative and litigation developments: See the state-by-state tracker on page 5 for current bill status (introduced, cleared, or passed).
- Model operational and financial impacts should contract-pharmacy participation narrow.
What to Expect Next
Contract-pharmacy access remains one of the most dynamic and unsettled areas of 340B policy. Covered entities should stay informed, coordinate with state associations, and proactively plan for potential changes in contract-pharmacy availability to preserve program stability and patient access.
Maine’s 340B Contract Pharmacy Law Clears Final Legal Hurdle
In late October, a federal court declined to block Maine’s 340B contract-pharmacy access law, allowing the statute to take full effect. Maine’s law requires drug manufacturers to provide 340B pricing for prescriptions dispensed through in-state contract pharmacies, even when national manufacturer policies restrict contract-pharmacy access. The ruling positions Maine among the growing number of states implementing enforceable protections for covered entities.
Key Provisions Moving Into Effect
Under the Maine statute, manufacturers now must:
- Honor 340B pricing for all covered outpatient drugs distributed or dispensed within Maine, regardless of contract-pharmacy designations.
- Refrain from conditioning 340B pricing on data reporting beyond what federal law mandates.
- Comply with state enforcement authority, including potential civil penalties imposed by Maine’s attorney general.
The ruling effectively clears the way for full enforcement, giving covered entities in Maine a more predictable contract-pharmacy environment.
Regional and National Momentum
The decision also adds to the emerging body of legal precedent supporting states’ authority to safeguard 340B contract-pharmacy access. For critical access hospitals, rural clinics, and health centers, Maine’s ruling preserves an essential access point for affordable medications. By allowing the law to stand, the court reinforced states’ ability to respond when manufacturer restrictions threaten reliable drug access.
What is especially notable about Maine’s development is the regional momentum it reflects. New England states—including New Hampshire and Vermont—have been closely evaluating similar statutory protections, and Maine’s outcome may encourage further legislative movement. This trend also underscores the broader dynamic of states filling policy gaps while federal 340B rulemaking remains limited.
What to Expect Next
Other states are monitoring the Maine ruling as they assess their own 340B contract-pharmacy frameworks. While each state’s approach varies, legislatures in New England, the Midwest, and the Pacific Northwest have shown increased interest in enacting measures that ensure manufacturer compliance with 340B pricing within their borders.
For multi-state covered entities, Maine’s decision serves as another indicator that contract-pharmacy compliance requirements will continue to differ across states. Updating internal compliance matrices and staying aware of state-specific obligations will be increasingly important.
Bottom Line
Maine’s contract-pharmacy law now advances without legal barriers, strengthening protections for covered entities and reinforcing a broader shift toward state-led 340B oversight. As more states consider similar measures, covered entities should remain attentive to developing legislative landscapes and prepare for an increasingly varied state-by-state compliance environment.
GS1 Releases First-Ever Guidance Focused on 340B Shipments
On November 12, GS1 (Global Standards 1) US released new guidance on how wholesalers, manufacturers, and contract pharmacies should track and document 340B shipments.
GS1 identifiers are not new, but this is the first time GS1 has issued recommendations specifically for 340B replenishment and drop-shipments, where product ownership and shipment routing can get confusing.
Why It Matters for Covered Entities
The biggest challenges in 340B replenishment usually involve unclear shipment records—for example, when a drug is drop-shipped to a contract pharmacy or when ship-to/ship-from locations don’t match what the TPA expects. These gaps cause issues in audits, replenishment validation, and now rebate submissions. GS1’s new guidance simply aims to standardize how supply-chain partners document these shipments, so covered entities have clearer, more reliable data.
Important: Covered entities do not need to create or manage GS1 identifiers themselves.
So What Does a Covered Entity Actually Need To Do?
Most 340B entities won’t make system changes. Instead, they should make sure the partners in their supply chain are aligned with the new guidance:
- Ask your wholesaler and TPA whether they support GS1 shipment data. This includes whether they can track basic identifiers and provide cleaner documentation for 340B shipments.
- Confirm how shipment information is collected for contract-pharmacy locations. Drop-shipments and mixed inventory often cause the most confusion—GS1’s guidance is meant to reduce those issues.
- Update expectations in contracts or service agreements when renewing. A simple line about supporting GS1-based shipment documentation can help ensure clean data going forward.
- Use the guidance as a checkpoint—not a compliance rule. GS1 is offering best practices, not requirements. Covered entities just need partners who can follow them.
What This Does Not Mean
- This is not a new federal rule.
- Covered entities do not need to change inventory systems.
- TPAs do not need to rebuild their platforms overnight.
- You will not be penalized for not using GS1 identifiers.
What to Expect Next
As the HRSA rebate model begins in 2026, shipment documentation will take on greater importance. GS1’s guidance is expected to influence how wholesalers, contract pharmacies, and TPAs standardize shipment data, helping ensure that what a CE purchased, received, and dispensed aligns cleanly with rebate submissions.
Covered entities won’t need to manage GS1 data themselves, but they should expect partners to strengthen documentation practices. This added clarity will support smoother rebate processing and improve audit readiness as data requirements increase. You can read more on these guidelines here.
Eliquis® WAC Reduction Announced
Bristol-Myers Squibb (BMS) will reduce the WAC price of Eliquis® beginning January 1, 2026—a change that may help ease some of the projected financial burden for covered entities anticipating significant impact from this product under the HRSA 340B Rebate Pilot. While the reduction will not eliminate the full estimated cost exposure, it should provide partial relief.
To support the transition, BMS is offering a brief rebate window for eligible purchases made December 22–31, 2025, with credits based on the difference between the old and new WAC prices. Entities planning inventory adjustments should finalize returns before December 1, as AmerisourceBergen will pause returns from December 1–January 5, and items returned after January 5 will be credited at the new, lower price.
Current Status on State Bills and Laws that Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates since last update
Industry Watch: PHRMA’s New Campaign
PhRMA has launched a new national advertising campaign titled “End Middlemen Markups. Put American Patients First.” The campaign claims that drug discounts and rebates often don’t reach patients and blames what it calls “middlemen markups” by hospitals, pharmacies, and other intermediaries. It specifically highlights hospitals participating in the 340B Drug Pricing Program, suggesting that some do not pass along their savings directly to patients.
The ads follow a series of similar PhRMA initiatives portraying the 340B program as lacking transparency. However, many covered entities maintain that 340B savings are reinvested in vital services like helping fund patient assistance programs, expand clinic access in rural areas, and sustain care for uninsured or underinsured populations.
What Covered Entities Should Know
While the campaign does not introduce new policy, it adds to a growing narrative around transparency and accountability in 340B operations. It’s important for covered entities to proactively demonstrate how program savings are used to improve access and offset uncompensated care. Doing so not only reinforces the integrity of the program but also provides valuable data should oversight efforts expand.
At the same time, the ad points to direct-to-patient delivery models as an example of how manufacturers say they’re “cutting out the middleman” to make medicines more affordable. The message positions drugmakers as problem-solvers in the pricing chain, contrasting them with hospitals and pharmacies portrayed as adding unnecessary markups. While these claims are primarily aimed at shaping public perception, they reflect a broader communications strategy to shift attention away from manufacturer pricing practices and onto the healthcare delivery system.
Action Steps for Covered Entities
- Document measurable patient benefit: Maintain clear, audit-ready records linking 340B savings to patient outcomes such as financial assistance, expanded service lines, or rural outreach.
- Monitor policy and advocacy developments: The ad campaign may shape legislative discussions; staying informed ensures your organization can respond with facts and data.
- Communicate your impact: Share stories and metrics showing how 340B directly supports your patients and community health mission.
Bottom Line
PhRMA’s latest campaign underscores a persistent focus on how 340B savings are used, but for covered entities, those savings remain a lifeline for patient access and community care. Strengthening documentation and communication around 340B’s patient impact is the best way to counter misconceptions, demonstrate transparency, and protect the program’s essential role in supporting America’s safety net.
Want every update delivered to your inbox? Subscribe to our newsletter at the bottom of our home page here

