This update focuses on rebate pilot preparedness, outlining recent legal and legislative activity, manufacturer participation trends, audit considerations, and the operational steps Covered Entities should be taking now.
Hospitals Challenge HRSA’s 340B Rebate Model Pilot
Opposition to HRSA’s proposed 340B Rebate Model Pilot has intensified as multiple groups move to halt implementation ahead of the program’s planned January 1, 2026 launch. In early December, hospitals, community-based providers, and national advocacy organizations pursued parallel legal and legislative efforts to stop or delay the pilot.
Hospitals File Federal Lawsuit
On December 1, 2025, hospital associations and safety-net providers filed a federal lawsuit seeking to block HRSA from implementing the 340B rebate pilot. The plaintiffs argue that HRSA lacks statutory authority to replace upfront 340B discounts with a rebate-based system and that the pilot imposes significant financial and administrative burdens on Covered Entities. Hospitals contend that requiring purchases at wholesale acquisition cost (WAC), even temporarily, threatens cash flow and could disrupt patient care, particularly for providers that rely heavily on 340B savings.
Providers Urge Congress to Pause Implementation
Days later, on December 5, more than 260 health care providers and advocacy organizations signed a letter urging congressional leaders to intervene. Organized by Advocates for Community Health (ACH), the letter asked House and Senate leadership to “take legislative action to pause implementation” of the rebate pilot.
Signatories included major national groups such as the National Association of Community Health Centers (NACHC) and the American Society of Health-System Pharmacists (ASHP), along with hundreds of individual providers and state and local associations. While many signers represented community health centers and other federal grantees, the concerns raised echoed those expressed by hospitals in their lawsuit.
A Growing Record of Opposition
These actions follow earlier efforts to stop the pilot, including:
- Strong opposition submitted to HHS during public comment periods
- A bipartisan October letter signed by 163 U.S. House members urging HRSA to abandon the pilot
- Public statements from provider groups warning of significant operational and financial strain
- Together, these developments reflect broad-based concern across the provider community and among lawmakers.
For Covered Entities, planning must account for multiple potential outcomes, including delay, modification, or partial rollout.
What to Expect Next: Essentials for January 1, 2026
- Ensure registration and access to the Beacon platform.
- Coordinate with third-party administrators and IT teams to update required data fields.
- Confirm internal and external ownership for rebate operations, including responsibility for submission, reconciliation, dispute management, and audit documentation.
- Engage your finance teams early to plan for temporary cash-flow impacts and ensure rebate receipts can be both tracked and posted accurately when payments begin.
Please visit HRSA’s 340B Rebate Model page for details on approved manufacturer plans, the rebate process, and implementation FAQs. You can also find additional insights in our previous ACI Monthly edition and 340B Rebate Model article.
Accelerating Court Activity Adds Complexity to 340B Planning
While the broader debate over HRSA’s 340B rebate pilot focuses on policy and statutory authority, a separate and equally important issue is how quickly the legal process is moving. With a January 1, 2026 implementation date approaching, courts have signaled that decisions affecting Covered Entity operations could come with little notice.
Court Sets Compressed Schedule
Following the December 1 hospital-led lawsuit, the court scheduled oral arguments for December 19, 2025, reflecting the potential for immediate financial and operational impact if the rebate pilot proceeds. The accelerated timeline increases the likelihood that hospitals may need to respond to legal outcomes within days rather than weeks.
For many organizations, this pace will limit their ability to gradually plan or test new workflows before they must adopt them.
Manufacturers Seek to Enter the Case
In early December, PhRMA and several drug manufacturers filed motions to intervene in the lawsuit. Their involvement does not change the pilot itself, but it does increase the complexity of the case and the range of outcomes the court may consider.
Manufacturer participation may lead to additional filings, requests for clarification, or appeals, extending uncertainty even if the court allows the pilot to move forward on schedule.
Why Speed Matters for Hospitals
Rapid legal developments create risk not because of the policy change itself, but because decisions may require action before systems, staffing, or financial planning can be fully adjusted. Short turnaround times increase the likelihood of last-minute guidance, shifting expectations, or uneven implementation across manufacturers and intermediaries.
In this environment, even well-prepared organizations may need to make operational decisions with incomplete information, increasing reliance on external partners and internal escalation.
Why It Matters for Covered Entities
The accelerated legal timeline increases the likelihood of:
- Last-minute operational guidance
- Short turnaround times for compliance changes
- Limited opportunity to test or phase in new processes
What to Expect Next
Court action following the December hearing may provide initial signals on whether implementation proceeds as scheduled, but it is unlikely to fully resolve the underlying legal questions. Additional motions, appeals, or requests for clarification could follow quickly, particularly if manufacturers remain actively involved.
As a result, Covered Entities should anticipate ongoing legal activity even after January 1, with the potential for evolving guidance during early implementation. Operational expectations may continue to shift as courts, HRSA, and manufacturers respond to early outcomes and practical challenges.
Varied Manufacturer Participation Shapes 340B Operations
HRSA’s 340B rebate pilot is now underway, but applied only to a defined set of drugs and manufacturers. This selective structure is creating uneven operational requirements across the 340B program and complicating how Covered Entities manage purchasing, billing, and reconciliation.
Impact Concentrated but Not Uniform
Under the current pilot design, only certain manufacturers and specific drugs qualify for rebate-based pricing. As a result, the operational impact differs across Covered Entities depending on which products they use, and which manufacturers supply them.
For many organizations, the practical impact concentrates on a relatively small number of drugs, though the specific products affected vary by Covered Entity. Those products may represent a significant portion of pharmacy spend or require more complex operational handling.
Parallel Workflows Now Required
Because the rebate pilot applies only to selected drugs, Covered Entities are often required to operate two pricing models at the same time:
- Traditional upfront 340B pricing for most products
- Rebate-based pricing for pilot-approved drugs
Managing parallel workflows increases complexity across purchasing, claims processing, data submission, and financial reconciliation. This environment also heightens the risk of errors, delays in realizing savings, and misalignment between policy and practice.
Why This Creates 340B Risk
The challenge is not simply administrative burden, but inconsistency. Different drugs may require different handling, different data flows, and different timing for realizing 340B savings. These distinctions must be tracked accurately and supported with documentation, particularly as HRSA audit expectations remain unchanged.
Without clear internal coordination, the selective nature of the pilot can introduce gaps between policy, system configuration, and day-to-day practice.
What to Expect Next
As the pilot continues, additional manufacturers or drugs may be added, but expansion is not expected to occur uniformly. Operational guidance may continue to evolve as HRSA, manufacturers, and intermediaries address early implementation challenges.
ACI has already shared preliminary, drug-specific impact estimates with its clients to help inform early planning. Covered Entities can use these preliminary estimates to prioritize internal planning discussions, validate cash-flow assumptions, and identify where additional operational support may be needed.
Bottom Line
The rebate pilot currently applies only to specific drugs and manufacturers, resulting in uneven application of rebate-based purchasing and reconciliation requirements across Covered Entities. Focusing attention on the subset of approved products can help organizations prioritize coordination and manage early operational variability.
HRSA Audits Continue Despite Program Uncertainty
As Covered Entities prepare for potential changes to how 340B pricing is delivered, HRSA has reinforced that audit activity and program integrity enforcement continue without pause. Recent updates to HRSA’s audit results underscore that compliance expectations remain firmly in place—even as operational complexity increases.
Audit Focus Areas Remain the Same, Execution Does Not
HRSA’s published audit results continue to highlight familiar findings such as diversion, duplicate discounts, contract pharmacy oversight, and documentation gaps. These core issues are not new, but the way they are evaluated may become more complicated as pricing and billing workflows evolve.
Rebate-based purchasing could increase auditing requirements by necessitating review of additional data points, reconciliation records, and supporting documentation to confirm that Covered Entities ultimately paid no more than the 340B ceiling price.
Why Audits May Become More Challenging
Periods of operational transition historically increase audit risk. New workflows, especially those involving delayed pricing adjustments, introduce more steps where errors, gaps, or misalignment between policy and practice can occur.
In a rebate environment, Covered Entities may need to demonstrate:
- Clear tracking of eligible purchases
- Accurate linkage between claims, invoices, and rebate receipts
- Documentation showing how and when pricing adjustments occurred
These expectations may be applied even if implementation guidance continues to evolve.
Why It Matters for Covered Entities
As transaction flows become more complex, audit readiness depends less on system sophistication and more on clarity and consistency. Incomplete documentation or unclear ownership of processes can increase exposure, even when intent is compliant.
Maintaining a clean audit trail may require closer coordination between pharmacy, billing, finance, and compliance functions.
What to Expect Next
HRSA is expected to continue routine audits throughout 2026 and may begin assessing rebate-related documentation once the pilot is operational. Early audits may focus on whether Covered Entities can clearly explain and support how rebate-based pricing was managed during the transition period.
Covered Entities should expect auditors to evaluate both outcomes and process controls, particularly during early implementation.
ACI continues to monitor regulatory developments and help its clients interpret and operationalize evolving guidance in a way that supports both compliance and audit readiness.
Bottom Line
The risk is not that audit rules are changing, but that operations are becoming more complex. In certain cases, this has included temporary limitations or pauses in participation for select drugs or entities while organizations finalize systems, data flows, and reconciliation processes.
Third-Party Administrator Readiness Varies Ahead of Rebate Pilot
As the January 1, 2026 launch of HRSA’s 340B Rebate Pilot approaches, some third-party administrators and contract pharmacy networks have begun adjusting operations in anticipation of rebate-based processing. In certain cases, this has included temporary limitations or pauses in participation for select drugs or entities while organizations finalize systems, data flows, and reconciliation processes.
While these measures aim to support accurate rebate administration, they may create short-term access or workflow changes for Covered Entities that rely on external pharmacy partners. Organizations should ensure they understand how their third-party administrators handle claims data submission, whether systems are fully prepared to process rebate-eligible drugs, and whether any temporary product or entity-level restrictions are in place.
Current Status on State Bills and Laws That Prohibit Drugmaker 340B Contract Pharmacy Restrictions
*States in Bold are the newest updates since last month
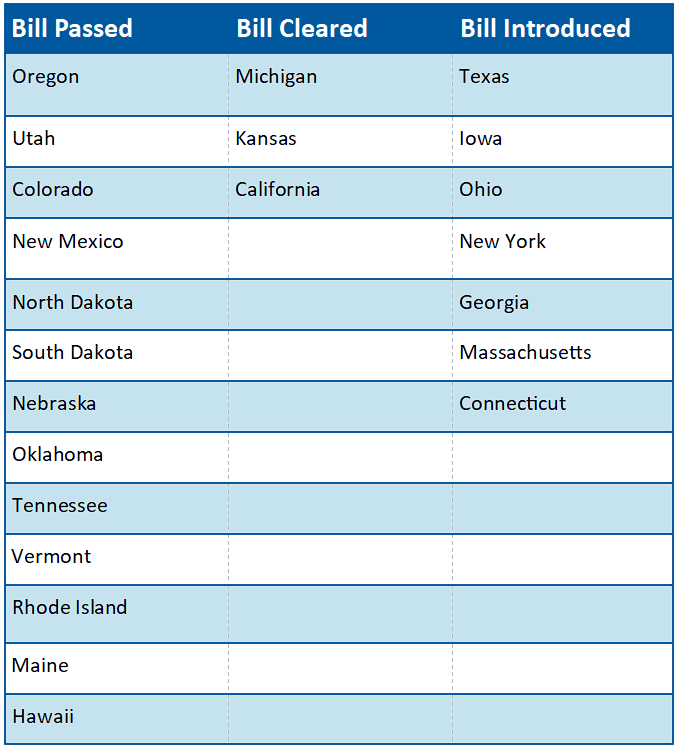
Contract Pharmacy Litigation Continues to Expand
Legal disputes over 340B contract pharmacy access continue to broaden, with manufacturers now challenging every state law enacted to protect Covered Entity use of contract pharmacies. Recent litigation highlights how these disputes are evolving beyond individual states and becoming a sustained national strategy.
New Mexico Lawsuit Highlights Expanding Legal Strategy
In December, the Pharmaceutical Research and Manufacturers of America (PhRMA) filed a federal lawsuit challenging New Mexico’s 340B contract pharmacy access law, which applies only to federally qualified health centers (FQHCs and look-alikes). The statute, set to take effect January 1, 2026, prohibits manufacturers from restricting contract pharmacy arrangements for health centers or requiring claims data beyond what federal law mandates.
PhRMA argues that federal statute preempts the law, that the law unlawfully regulates interstate commerce, and that it is unconstitutionally vague. While narrower in scope than other state laws, the challenge signals that manufacturers are unwilling to concede even limited state-level protections.
A National Litigation Strategy Takes Shape
With the New Mexico filing, manufacturers have now brought federal lawsuits challenging all 21 states that have enacted 340B contract pharmacy access laws. Outcomes to date have varied by jurisdiction, with several courts upholding state protections while others have issued temporary injunctions or allowed challenges to proceed.
Rather than converging toward a single resolution, these cases suggest that state-specific rulings and ongoing appeals will continue to shape contract pharmacy access, creating a fragmented legal environment.
Why It Matters for Covered Entities
Contract pharmacy access often underpins patient reach and program sustainability. Even when litigation targets specific entity types, court rulings and manufacturer responses can influence broader policy decisions, including shipment practices, data requirements, and participation conditions.
Because many Covered Entities rely on a limited number of pharmacy partners, changes driven by litigation can have immediate operational and financial consequences.
What to Expect Next
Courts will continue issuing rulings on state contract pharmacy laws throughout 2026, with appeals likely in several cases. No single decision is expected to resolve this issue nationwide.
Covered Entities should anticipate continued variation by state and manufacturers, rather than uniform standards, and monitor how legal outcomes translate into operational policy changes.

